Reactors: Difference between revisions
| Line 37: | Line 37: | ||
=General Reactor Design= | =General Reactor Design= | ||
The design of the reactor should not be carried out separately from the overall process design due to the significant impact on capital and operating costs on other parts of the process[1]. | |||
==Collect Required Data== | ==Collect Required Data== | ||
Out of all process equipment, reactor design requires the most process input data: reaction enthalpies, phase-equilibrium constants, heat and mass transfer coefficients, as well as reaction rate constants. All of the aforementioned parameters can be estimated using simulation models or literature correlations except for reaction rate constant constants, which need to be determined experimentally. | Out of all process equipment, reactor design requires the most process input data: reaction enthalpies, phase-equilibrium constants, heat and mass transfer coefficients, as well as reaction rate constants. All of the aforementioned parameters can be estimated using simulation models or literature correlations except for reaction rate constant constants, which need to be determined experimentally. | ||
===Enthalpy of Reaction=== | ===Enthalpy of Reaction=== | ||
The heat given out in a chemical reaction is based on the enthalpies of the component chemical reactions, which are given for standard temperature and pressure (1 atm, 25 C). | |||
===Equilibrium Constant and Gibbs Free Energy=== | ===Equilibrium Constant and Gibbs Free Energy=== | ||
Revision as of 04:35, 7 February 2014
Title: Reactors
Author: Sean Cabaniss, David Park, Maxim Slivinsky and Julianne Wagoner
Steward: Fengqi You
Date Presented: February 4, 2014 /Date Revised: February 4, 2014
Introduction
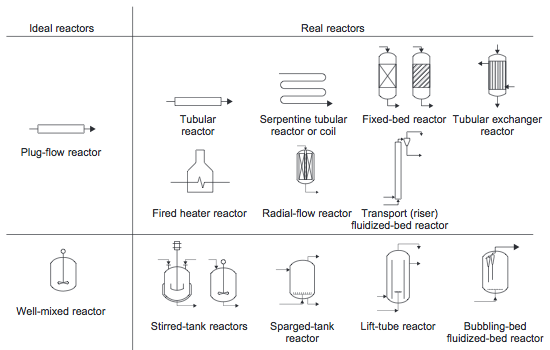
Ideal Reactors
Plug Flow Reactor (PFR)
A PFR with tubular geometry has perfect radial mixing but no axial mixing. All materials hav the same residence time, τ, and experience the same temperature and concentration profiles along the reactor. Equation for PFR is given by:
where M = molar flow rate, dV is the incremental volume, and R is the rate of reaction per unit volume.
This equation can be integrated along the length of the reactor to yield relationships between reactor resident time and concentration or conversion.
Continuously Stirred Tank Reactor (CSTR)
In a CSTR there is no spatial variation- the entire vessel contents is at the same temperature, pressure, and concentration. Therefore the fluid leaving the reactor is at the same temperature and concentration as the fluid inside the reactor.
The material balance across the CSTR is given by:
General Reactor Design
The design of the reactor should not be carried out separately from the overall process design due to the significant impact on capital and operating costs on other parts of the process[1].
Collect Required Data
Out of all process equipment, reactor design requires the most process input data: reaction enthalpies, phase-equilibrium constants, heat and mass transfer coefficients, as well as reaction rate constants. All of the aforementioned parameters can be estimated using simulation models or literature correlations except for reaction rate constant constants, which need to be determined experimentally.
Enthalpy of Reaction
The heat given out in a chemical reaction is based on the enthalpies of the component chemical reactions, which are given for standard temperature and pressure (1 atm, 25 C).
Equilibrium Constant and Gibbs Free Energy
Reaction Mechanisms, Rate Equations, and Rate Constants
Heat and Mass Transfer Properties
Select Reaction Conditions
Chemical or Biochemical Reaction
Catalyst
Temperature
Pressure
Reaction Phase
Solvent
Concentrations
Determine Materials of Construction
Determine Rate-Limiting Step and Critical Sizing Parameters
Mixing
Heating and Cooling
Basic Principles
Stirred Tank Reactors
Catalytic Reactors
Preliminary Sizing, Layout, and Costing of Reactor
Estimate the Performance
Optimize the Design
Multiphase Reactors
Reactor Design for Catalytic Processes
Design of Bioreactors
Safety Considerations in Reactor Design
Capital Cost of Reactors