Design S2: Difference between revisions
| (3 intermediate revisions by the same user not shown) | |||
| Line 39: | Line 39: | ||
===Reactor=== | ===Reactor=== | ||
The hydrogenation reaction occurs inside of the reactor thanks to the packed catalyst bed. The catalyst used is 0.4% Fe, 1.9% Na, 2.66% Ag, 2.66% Pd, 10.0% Re on 1.5mm carbon support. With this catalyst, BDO is produced with over 90% selectivity and minimal side reactions of THF and GBL | The hydrogenation reaction occurs inside of the reactor thanks to the packed catalyst bed. The catalyst used is 0.4% Fe, 1.9% Na, 2.66% Ag, 2.66% Pd, 10.0% Re on 1.5mm carbon support. With this catalyst, BDO is produced with over 90% selectivity and minimal side reactions of THF and GBL (Bhattacharyya and Manila, 2011). The reaction has an operating pressure of 2000-4000 psi and internal reactor temperature of 165°C. This temperature allows for about 99.7% conversion of succinic acid (Bhattacharyya and Manila, 2011). Due to the exothermic nature of the reaction, a cooling jacket is required which utilizes downstream cold streams to cool the internal bed to maintain the desired reaction temperature. | ||
===Post-reactor=== | ===Post-reactor=== | ||
| Line 89: | Line 89: | ||
==Economics== | ==Economics== | ||
===Equipment costing=== | ===Equipment costing=== | ||
The calculated capital costs from Aspen Economic Analyzer are reported in Table 2 for all process equipment. The size of the reactor was calculated from the liquid hourly space velocity given in the 2011 ISP patent | The calculated capital costs from Aspen Economic Analyzer are reported in Table 2 for all process equipment. The size of the reactor was calculated from the liquid hourly space velocity given in the 2011 ISP patent (Bhattacharyya and Manila, 2011) and a void fraction estimate of 0.4. Storage tanks were sized to contain up to two continuous weeks of material. The number of distillation trays in each tower and the flow rates through pumps, vessels, and the flare were calculated in HYSYS. Heat exchange areas were given in Aspen Energy Analyzer or from HYSYS. | ||
[[File:Equipment_cost.JPG|center|]] | [[File:Equipment_cost.JPG|center|]] | ||
| Line 131: | Line 131: | ||
==References== | ==References== | ||
Bhattacharyya A, Manila MD, inventor; ISP Investments Inc., assignee. Catalysts for maleic acid hydrogenation to 1,4-butanediol. United States Patent US 7935834 B2. 2011 May 3. | |||
Burk MJ, Stephen J, Dien SJV, Burgard AP, Niu W, inventor; Genomatica Inc., assignee. Compositions and methods for the biosynthesis of 1,4-butanediol and its precursors. United States Patent US8067214 B2. 2011 Nov 29. | |||
Chung SH, Kim MS, Eom HJ, Lee KY. Hydrogenation of Succinic Acid Using Ruthenium Nanoparticles Embedded Catalysts. Proceedings of 2013 AIChE Annual Meeting; 2013 Nov 6; San Francisco, USA. | |||
Icis.com. Chemical industry awaits for bio-succinic acid potential [Internet]. Surrey: Reed Business Information Limited; c2015 [cited 2015 Feb 26]. Available from: http://www.icis.com/resources/news/2012/01/30/9527521/chemical-industry-awaits-for-bio-succinic-acid-potential/. | |||
Ingram A, Le B. 1,4-butanediol/tetrahydrofuran (BDO/THF) [Internet]. Wheaton: Nexant Inc.; c2011- [updated 2013 Apr; cited 2015 Feb 28]. Available from: http://thinking.nexant.com/sites/default/files/report/field_attachment_abstract/201304/2012_3_abs.pdf. | |||
Nexant.com. Is Bio-Butanediol Here to Stay [Internet]? Wheaton: Nexant Inc.; c2000–15 [cited 2015 Feb 28]. Available from: http://www.nexant.com/about/news/bio-butanediol-here-stay. | |||
Nicnas.gov.au. Butanediol (1,4-butanediol) factsheet [Internet]. Sydney: National Industrial Chemicals Notification and Assessment Scheme [cited 2015 Feb 28]. Available from: http://www.nicnas.gov.au/communications/publications/information-sheets/existing-chemical-info-sheets/other-information-sheets. | |||
Orbichem.com. Chemical Market Insight & Foresight-On A Single Page 1,4-Butanediol [Internet]. Tecnon OrbiChem; c2004-15 [cited 2015 Feb 26]. Available from: http://www.orbichem.com/userfiles/CNF%20Samples/bdo_13_11.pdf. | |||
Latest revision as of 22:43, 28 February 2015
Title: Production of 1,4-butanediol
Authors: Nick Pinkerton, Karen Schmidt, and James Xamplas
Date Presented: March 14, 2014
Introduction
1,4-butanediol (BDO) is traditionally made from petroleum-derived feedstocks in a variety of processes such as the Reppe (acetylene-based), Mitsubishi (butadiene-based), and Davy (maleic acid-based) processes (Nexant.com). Recently, because of the continually high price of crude oil and the desire to be environmentally-conscious, there has been a push toward the use of feedstocks derived from biomass. Bio-succinic acid can be easily used in the Davy process as a substitute for maleic acid to form the end product, BDO (Nexant.com). It is this process that Team S2Kool4Skool has chosen to develop for a new bio-butanediol plant, because the Davy method is mature and requires no new innovations, and because an appropriate feedstock of bio-succinic acid is now available. The plant will be located in East Carroll Parish, Louisiana. This county is also home to Myriant; located in Lake Providence, they are the largest domestic provider of bio-succinic acid (Icis.com). Furthermore, Lake Providence is conveniently located on the Mississippi River, which will allow for affordable transportation of our bio-based butanediol product. The intent of this project is to produce 45,000 metric tonnes of 99.5 wt% 1,4-butanediol per year in a new plant in Lake Providence, LA. The plant will run 24 hours per day for 350 days out of the year, allowing approximately two weeks for a maintenance shutdown.
As there are several techniques for producing 1,4 butanediol in industry, the first step was to determine a synthesis path. Based on the chosen path, reaction kinetics, and required production rate of the process, a reactor system was then designed. Next, the process was designed to deliver the reactants to the reactor at the proper operating conditions, and the separations, purification, and waste management steps were designed. Cost estimates were made using costing software and hand calculations. Finally, the process was iterated and optimized to reduce costs.
Background
General information
BDO is an organic chemical with the molecular formula . It is also a diol with its two hydroxyl groups located at the terminal carbons. BDO has a boiling point of 235 degrees C and is therefore a colorless liquid at standard temperatures and pressures (Nicnas.gov.au).
There are many precursors that petrochemical synthesis of BDO uses. The commonality between all of the precursor acids is that they are hydrogenatable. In replacement of one of these petrochemical precursors, our team has been tasked with the challenge of deriving BDO from the bio-based precursor succinic acid. This catalyzed reaction will take place in the presence of hydrogen as the hydrogenating component shown below:
As described in the above reaction, there will need to be at least 4:1 stoichiometric ratio of hydrogen gas to succinic acid. Two moles of water will also be produced along with each mole of BDO. This reaction is exothermic, which requires the reactor to be continuously cooled to maintain our reactor temperature. There are also side products that are produced which include tetrahydrofuran (THF) and γ-butyrolactone (GBL); however, thanks to catalyst selectivity these byproducts are produced in small quantities.
Market analysis
Butanediol has a quickly expanding market due to new technological evolutions and its growing use as a chemical intermediary in advanced materials. With biological routes being optimized, the potential of biomass-derived chemicals is tremendous. The global demand of BDO was estimated at 1.5 million metric tons in 2011 and is projected to grow at an annual rate of 4.5% for the next several years (Nexant.com). Unlike other chemical products, BDO’s profitability and attractiveness to producers lies in its downstream potential. Figure 1 below demonstrates several potential downstream products that can be directly or indirectly synthesized from BDO. The largest of these contributors include THF and GBL.
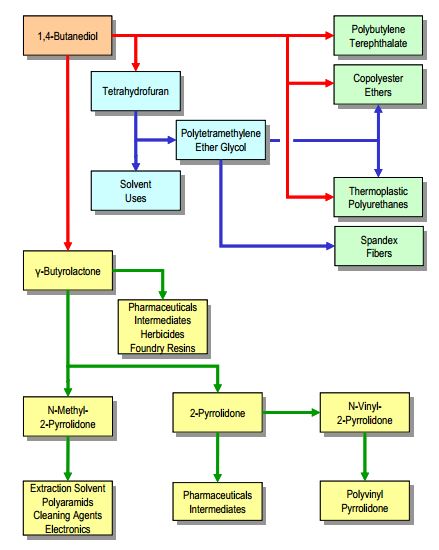
The current market demand of BDO is being supplemented by several global chemical companies. These companies include BASF, BioAmber, Purac, Myriant, DSM, Mitsubishi Chemical, Roquette, and OPXBIO. The market shares of these companies were not available for this product; however, with the consistent growth of the BDO market our team feels that the market is not saturated or impenetrable. The current market price of BDO fluctuates between $3.06 and $3.31 per kg for US-made products (Orbichem.com). These prices correlate to a nearly 7 billion dollar industry.
Process alternatives
1,4-butanediol is traditionally made from petroleum-derived feedstocks in a variety of processes (Ingram and Le, 2013). Recently, because of the continually high price of crude oil and the desire to be environmentally-conscious, there has been a push toward the use of feedstocks derived from biomass. Several companies are currently implementing bio-routes of producing butanediol. Genomatica is using a bioengineered microorganism to convert sugar feedstocks directly to BDO via fermentation (Burk et al., 2011), but most companies are instead using microorganisms to convert sugar to succinic acid. The bio-succinic acid can be easily used in the Davy process as a substitute for maleic acid to form the end product, which is the path that most are choosing, although research is being conducted on alternative pathways (Chung et al., 2013).
Process overview
The newly proposed plant can be divided into four stages: pre-reactor, reactor, post-reactor and distillation. Each section is an integral part to the overall process and demands close attention. See the complete process flow diagram in Figure 2.
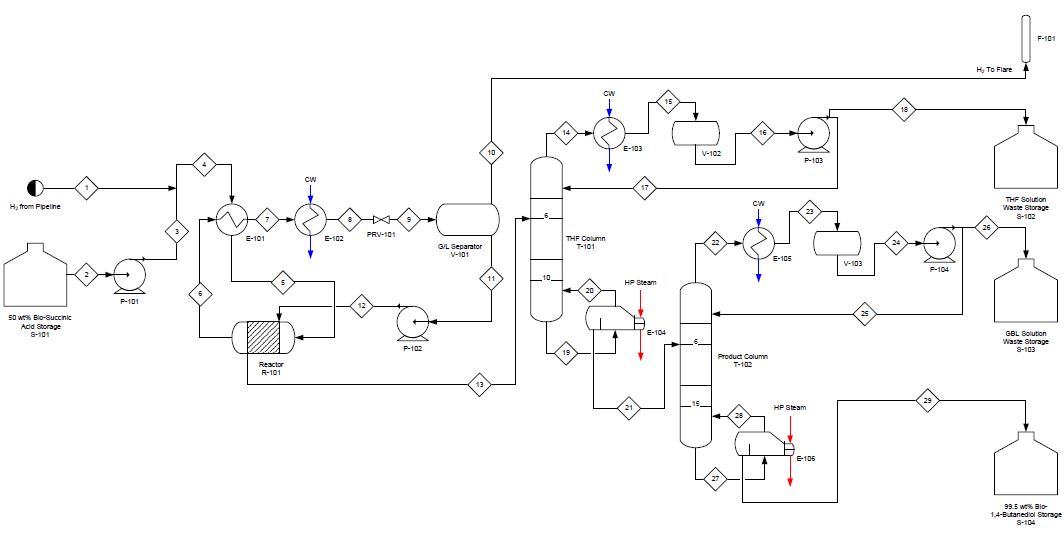
Pre-reactor
This chemical process begins with two feedstocks: hydrogen gas and bio-succinic acid. The hydrogen gas will be obtained from a pipeline at 150 atm and will be used in molar excess inside the reactor. The bio-succinic acid will be purchased by Myriant Corporation which is located near the planned plant site. The pricing for this feedstock is approximately $2.12 per kg. The plant will require 63,000 metric tonnes of bio-succinic acid per year to meet the proposed plant capacity. First the bio-succinic acid feed is pumped up to 150 atm to match the hydrogen gas feed, and then it is mixed with the hydrogen gas prior to being sent into heat exchanger E-101 for heating. The heat exchanger brings the two feeds up to 110oC and sends them to the jacketed packed bed reactor.
Reactor
The hydrogenation reaction occurs inside of the reactor thanks to the packed catalyst bed. The catalyst used is 0.4% Fe, 1.9% Na, 2.66% Ag, 2.66% Pd, 10.0% Re on 1.5mm carbon support. With this catalyst, BDO is produced with over 90% selectivity and minimal side reactions of THF and GBL (Bhattacharyya and Manila, 2011). The reaction has an operating pressure of 2000-4000 psi and internal reactor temperature of 165°C. This temperature allows for about 99.7% conversion of succinic acid (Bhattacharyya and Manila, 2011). Due to the exothermic nature of the reaction, a cooling jacket is required which utilizes downstream cold streams to cool the internal bed to maintain the desired reaction temperature.
Post-reactor
The effluent of the reactor is sent back to E-101 as the hot stream. After exiting E-101, the reactor product stream is sent to a secondary exchanger, E-102, where utility cooling water is used to reduce the temperature to an acceptable temperature prior to sending it to a pressure let-down valve. At this point the pressure of the stream is taken from 150 atm to 1 atm. This large pressure drop allows for the stream to split into its vapor and liquid portions in a gas-liquid separator. The vapor stream of the gas liquid separator is primarily hydrogen gas and sent to a flare for disposal. The liquid effluent is at approximately 45oC leaving the separator and is therefore pumped to the reactor jacket for the reaction cooling mentioned previously. After running through the reactor jacket, the stream enters the separation processes.
Distillation
The first distillation column, T-101, is a 10 stage column whose primary purpose is separating the THF from the product feed. Due to THF’s lower boiling point, the byproduct comes off of the top of the column with mostly water. This distillate is sent to the plant's THF waste storage tank that has the capacity of two weeks. The bottoms of the column is sent to the subsequent distillation column that separates the BDO from the GBL and water. The relatively close boiling points of BDO and GBL, 235oC and 204oC, respectively, create a difficult separation that requires a 15 stage column. The distillate of the column is approximately 23% GBL with balance water. This stream is sent to a storage tank with similar sizing parameters as the THF storage tank. The bottom stream is the final 99.5wt% BDO product. This stream is sent to the final product tank, S-104. Depending on our customer demands and the plant location we have the ability to barge, rail or pipe our product to its final destination. Due to low purity of the byproducts, future iterations are needed to optimize either purifying byproducts or selling impure byproducts. There is definitely an available market for these byproducts that should be researched more extensively to increase profit.
Mass and energy balances
Using an Aspen HYSYS simulation we were able to record the material and energy streams going in and out of the process system. As expected, the material and energy totals for the inlet and outlet streams add together to equal 0. This proves that our system is mathematically prudent and thermodynamically feasible. The total mass flow of the system is 15,639 kg/h and the total energy in and out of the system is 1.41e8 kJ/h.
HYSYS simulation
The HYSYS simulation was performed using the NRTL ideal fluid package. After using AspenPlus to verify that the HYSYS package had the appropriate vapor-liquid equilibrium information between THF and water, and between GBL and water, we concluded it was feasible to proceed with that fluid package. The simulation consists of a reactor, a gas-liquid separator, 2 pumps, 3 heaters, a valve, and 2 distillation columns (see Figure 3). The simulation successfully converted the succinic acid feed into the desired products. Also, the combination of the two distillation columns was able to effectively separate the BDO to obtain a 99.5% pure product with 99.5% recovery. In addition, a set was made between the energy required to heat stream 12 and the energy required to cool the reactor so that these values were made equal. Lastly, the condenser and reboiler duties were used in four heat exchangers in order to determine the appropriate size of this equipment as well as the necessary utility flow rates.

Health and safety
Chemical properties
Inherent to this process are a number of toxic chemicals. Table 1 summarizes the important safety data including hazard type, odor, color, and exposure limits.
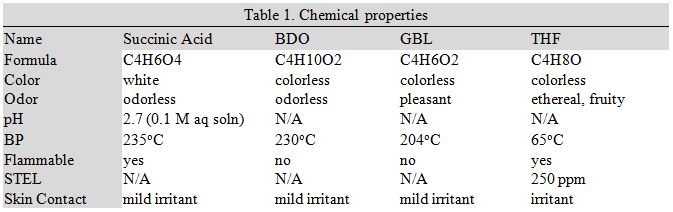
As shown in the above table, the chemicals that this facility will be dealing with will be relatively mild and non-life threatening. Regardless of their perceived threat, chemicals should always be handled with care especially when they are at high temperatures and pressures.
Safety procedures
Fire. There are many flammable materials that will be included in this process; therefore, fire safety is imperative for all employees. There are countless possible causes of ignition and care should be taken while handling any flammable material whether in the lab or in the field.
In case a fire is present on, the following protocol should be implemented:
- Small fire: Use DRY chemical powder.
- Large fire: Use alcohol foam, water spray, or fog.
- Call for backup if unable to control.
- Immediately contact supervisor and emergency personnel on site.
- Evacuate to safe distance in case of fires around any hazardous materials or pressure vessels.
Spills. Process chemical spills will eventually occur in a plant of this scale. Small spills are likely to occur in a laboratory setting. Large spills could be a result of loss of containment in the system. We must ensure that all personnel are aware of proper spill mitigation protocol.
- Small spill: Dilute with water and mop or absorb with inert dry material. Dispose of in proper receptacle.
- Large spill: Keep away from sources of ignition and heat. Prevent rundown to any drains or sewers. Call for assistance on disposal. Absorb material with DRY earth or other non-combustible materials.
- If spill is due to a loss of containment in the system, quickly consult the PLC and shut any valves to prevent further loss.
Exposure. We must ensure that our employees are aware of the possible toxicity levels of each substance and how to handle exposure. The chemicals that are being used in this process are known chemical irritants to the eyes, skin, and throat. Safety measures must be in place to acknowledge this hazard. Due to the possibility of high pressure releases, we will have 2-minute emergency oxygen masks placed strategically throughout the plant to ensure the safety of any operator in the presence of a large release.
If exposed to the process chemicals, find nearest eye wash station or safety shower immediately and flush exposed skin for at least 15 minutes. Remove any contaminated clothing. Seek medical attention immediately.
Storage. Store chemicals in segregated and approved areas. Any closed containers for laboratory purposes should be placed in cool, well-ventilated areas.
Economics
Equipment costing
The calculated capital costs from Aspen Economic Analyzer are reported in Table 2 for all process equipment. The size of the reactor was calculated from the liquid hourly space velocity given in the 2011 ISP patent (Bhattacharyya and Manila, 2011) and a void fraction estimate of 0.4. Storage tanks were sized to contain up to two continuous weeks of material. The number of distillation trays in each tower and the flow rates through pumps, vessels, and the flare were calculated in HYSYS. Heat exchange areas were given in Aspen Energy Analyzer or from HYSYS.
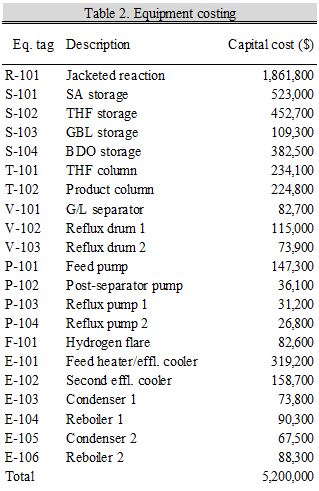
In addition to the above equipment, our plant will require an ion exchanger to produce deionized process water from the municipal water. This is estimated to cost $42,000 in capital cost.
Cash flow analysis
Summarizing the important takeaways from the economic analysis, this process will return a revenue of 144 MM$ annually. Offset by production costs, the yearly cash flow is approximately 2.7 MM$, except for in the years in which the catalyst must be reinstalled (approximately every 5 years). At a cost of 2.5 MM$, the cash flows in those years decrease to approximately 0.2 MM$. This analysis assumes the plant will take 2 years to construct, and will operate at 50% in its first year. Furthermore, an interest rate of 10% was assumed with a tax rate of 38% (the maximum for corporate gains taxes). Using the 7-year MACRS depreciation method, the 20 year NPV for the project is 4.3 MM$. With an IRR of 15.1%, which is greater than the assumed interest rate, this project looks to be profitable. Further optimization techniques should be used in future iterations to further increase profitability. See Tables 3 and 4 for key economic information.
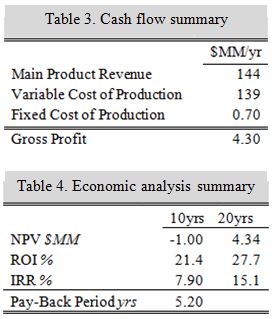
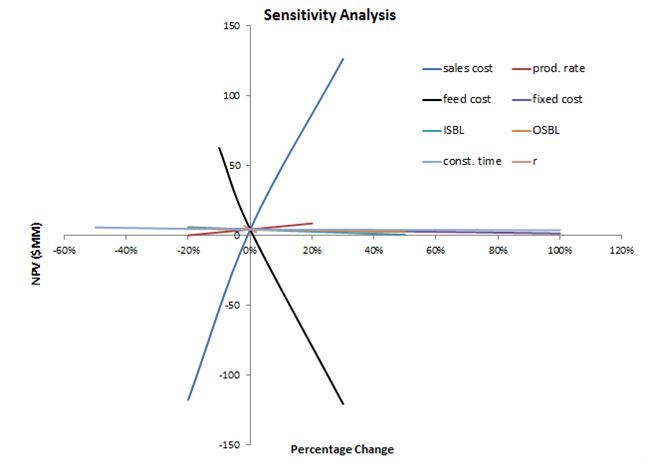
Sensitivity analysis
As seen in Figure 4, the economic evaluation of this process is influenced the most by changes in the sales price and feed cost. A 20% increase in sales price will result in an approximate 120 MM$ increase in NPV whereas a decrease of the same percentage will result in over a 120 MM$ decrease in NPV. Inversely, a 20% increase in feed cost will result in a 115 MM$ decrease in NPV and a 20% decrease in feed cost will result in a 60 MM$ increase in NPV.
To alleviate the risk associated with feed price, the team researched the price forecasting of bio-succinic acid. We compared the bio-succinic acid price to the adipic acid price over the six years between 2006 and 2012. The price of bio-succinic has remained relatively stable over this time period. Adipic acid, which is a common petrochemical precursor for BDO production, has had large fluctuations in price that result in unstable cash flows and uncertainty from shareholders. Thankfully, the stability of bio-succinic acid is a good sign that this process has a huge potential to be profitable especially if the demand for BDO continues to rise as projected.
Discussion
As described previously, the Davy process was selected as the most promising process of BDO production. The modified Davy process involves the hydrogenation of bio-succinicic acid with a Pa,Ag,Re catalyst and hydrogen gas. This process has many advantages, one being that it has mature technology that has been improved over the last 20 years. Additionally, the conversion to of succinic acid is virtually complete. Finally, overall plant yields can reach as high as 94 mol%.
Streams ahead of the pressure valve (PRV-101) are at approximately 1.51e4 kPa, and streams after the pressure valve are between 100 and 450 kPa. Furthermore, operating temperatures do not exceed 200°C, including safety factors. These operating conditions contribute to the feasibility of the process, as all the components can be designed with reasonable dimensions (wall thickness, cap type, etc.).
Of particular interest are the large costs of the reactor and pump P-101. The reactor costs nearly 2 MM$ because it has a moderate size with a liquid volume of 16.7 m3, and it operates at 150 atm, which requires a strong stainless steel shell for safety purposes. Pump P-101 has such a large capital cost because it is a centrifugal multistage pump that also needs to be made of stainless steel to withstand pressuring the succinic acid feed to 150 atm.
Financial indicators for the proposed plant suggest that it will be moderately profitable, and additional optimization should be performed before making the capital investment. There is a fair amount of risk associated with the implementation of this production facility, and the return at this juncture may not justify the risk.
The process design and simulation in HYSYS has relied on several key assumptions, which are cause for certain limitations to the results. First, the conversions and selectivity of the catalyst, while taken from literature sources, are assumed to be true. Furthermore, the assumption of negligible side reactions and products was made. It is possible that small waste products in the form of succinates also form in the reactor. Therefore, the main limitation of this model is generally due to reaction specifications.
There are certain safety concerns that go along with the proposed process. First, there are many pipes and vessels that will be at a pressure of approximately 150 atm. Extra insulation and protection must be applied to the piping that contains highly pressurized fluids, to prevent operator injury. Furthermore, the PFR is at a slight risk for runaway reactions due to the exothermic nature of the reaction. The temperature of the reactor must always be monitored by an operator, and the jacket cooling system requires a backup system as well. A benefit of this process is that there are no toxic or severely hazardous components in the process. Additionally, there are no instances of temperature greater than 200°C.
The process design will be fitted with instrumentation and controls to ensure stable operation. These controls, including sensors and valves, minimize potential damage to components due to variation in plant conditions, as well as optimizing the overall performance. For example, the piping entering the tower will be fitted with a control loop. In the direction of flow, a sensing instrument first detects the pressure of the fluid, and then sends a signal to the controller. If the pressure is not within the operating limits, the actuator is signaled to close or open a control valve which is located farther down the stream.
Conclusion
The Evanston Chemical Engineering Division suggests a process that produces 45,000 tonnes/year of 99.5 wt% 1,4-butandeiol to enter the market with an appropriate market share. The feasibility of producing BDO as a significant process was investigated. A wide range of BDO production processes were researched, with a modified Davy process being identified as having the most potential. Using a Pa,Ag,Re catalyst on carbon support, this process converts bio-succinic acid to BDO with approximately 94% conversion. This process was designed in Aspen HYSYS, and economic analysis was conducted in Aspen Economic Analyzer. Total capital costs of the project are 12.2 MM$, with a simple payback period of 5.2 years. Total operational costs annually are 139.7 MM$. The process design was successful, with the capability of producing the desired production of BDO.
Recommendations
Given the moderate profitability of this process design, it is recommended that Evanston Chemicals explores further optimization techniques to increase the profitability of this process. This includes investigating sales of the γ-butyrolactone byproduct, recycling process water, and further optimizing utility streams for the distillation tower.
Second, we would like to investigate recycling the 2300 kg/h of water produced in the reaction. Our succinic acid enters the reactor as a 50 wt% solution in water, so we should look into replacing some of this with the product water. Not only will this decrease our water bill, but it will also lower the cost of ion exchange.
References
Bhattacharyya A, Manila MD, inventor; ISP Investments Inc., assignee. Catalysts for maleic acid hydrogenation to 1,4-butanediol. United States Patent US 7935834 B2. 2011 May 3.
Burk MJ, Stephen J, Dien SJV, Burgard AP, Niu W, inventor; Genomatica Inc., assignee. Compositions and methods for the biosynthesis of 1,4-butanediol and its precursors. United States Patent US8067214 B2. 2011 Nov 29.
Chung SH, Kim MS, Eom HJ, Lee KY. Hydrogenation of Succinic Acid Using Ruthenium Nanoparticles Embedded Catalysts. Proceedings of 2013 AIChE Annual Meeting; 2013 Nov 6; San Francisco, USA.
Icis.com. Chemical industry awaits for bio-succinic acid potential [Internet]. Surrey: Reed Business Information Limited; c2015 [cited 2015 Feb 26]. Available from: http://www.icis.com/resources/news/2012/01/30/9527521/chemical-industry-awaits-for-bio-succinic-acid-potential/.
Ingram A, Le B. 1,4-butanediol/tetrahydrofuran (BDO/THF) [Internet]. Wheaton: Nexant Inc.; c2011- [updated 2013 Apr; cited 2015 Feb 28]. Available from: http://thinking.nexant.com/sites/default/files/report/field_attachment_abstract/201304/2012_3_abs.pdf.
Nexant.com. Is Bio-Butanediol Here to Stay [Internet]? Wheaton: Nexant Inc.; c2000–15 [cited 2015 Feb 28]. Available from: http://www.nexant.com/about/news/bio-butanediol-here-stay.
Nicnas.gov.au. Butanediol (1,4-butanediol) factsheet [Internet]. Sydney: National Industrial Chemicals Notification and Assessment Scheme [cited 2015 Feb 28]. Available from: http://www.nicnas.gov.au/communications/publications/information-sheets/existing-chemical-info-sheets/other-information-sheets.
Orbichem.com. Chemical Market Insight & Foresight-On A Single Page 1,4-Butanediol [Internet]. Tecnon OrbiChem; c2004-15 [cited 2015 Feb 26]. Available from: http://www.orbichem.com/userfiles/CNF%20Samples/bdo_13_11.pdf.