Desalination - Team B: Difference between revisions
Cindyxchen (talk | contribs) No edit summary |
Cindyxchen (talk | contribs) |
||
| (26 intermediate revisions by the same user not shown) | |||
| Line 1: | Line 1: | ||
Reverse Osmosis Desalination Plant in Durban, South Africa | |||
Authors: Lauren Burke, Cindy Chen, Osman Jamil, Natalia Majewska | Authors: Lauren Burke, Cindy Chen, Osman Jamil, Natalia Majewska | ||
| Line 9: | Line 9: | ||
==Executive Summary== | ==Executive Summary== | ||
An imminent global shortage of water will greatly affect the lives of millions of people. The situation in South Africa is predicted to exacerbate in the upcoming decades. This growing need for fresh drinking water motivated the design of a reverse osmosis desalination plant in the suburbs of Durban on the eastern coast of South Africa. A reverse osmosis process was selected based on the water salinity in the area and recent technological advancements in membrane efficiency. | |||
The process is designed to operate for 350 days a year, 24 hours a day to yield 132,000 m3 of water per day. Prior to reverse osmosis, the seawater is pretreated to prevent membrane fouling using a cascade of filters and high-pressure pumps. Reverse osmosis is carried out at 37 bar with two banks containing 900 and 4000 membranes, respectively. After reverse osmosis, the pressurized concentrate that contains the unwanted salts and ions is sent to an energy recovery device to pressurize the reverse osmosis feed. The deionized water from the reverse osmosis system is remineralized and disinfected before being sent to the consumer. | |||
Based on a thorough economic analysis, it has been determined that the process is not feasible at this stage. The annual estimated revenue is $37.5 MM. In addition, the 20 year NPV of the plant is -$23.5 MM with an IRR of 2.9%. A sensitivity analysis indicated that an increase in the price of water could potentially increase profits. Therefore, the design may become profitable implemented with the aid of government subsidies or if a drastic increase in water scarcity increases the selling price of water. | |||
__TOC__ | __TOC__ | ||
| Line 15: | Line 20: | ||
==Background== | ==Background== | ||
Close to 1.2 billion people, approximately one fifth of the world’s population, reside in areas of physical water scarcity [1]. Although there is enough water in the world for a population of seven billion people, it is unevenly distributed, and many regions have been experiencing water shortages for more than a decade. Given the current rate of population growth and water consumption, many studies predict that this problem will be exacerbated in the years to come. Water desalination is a process used to purify water so that it meets drinking standards for consumption. The two main technologies used for desalination are thermal desalination and reverse osmosis. While thermal desalination is a robust method that scales well, it is typically preferable for inlets of very high salt concentrations and very low energy costs, such as the gulf coast region. Reverse osmosis plants are more prevalent elsewhere, making up 80% of all desalination plants in the world [2]. Given the salinity of the inlet flow for this process and advances in technology, reverse osmosis has been selected as the optimal choice for this project. | |||
==Location Selection== | ==Location Selection== | ||
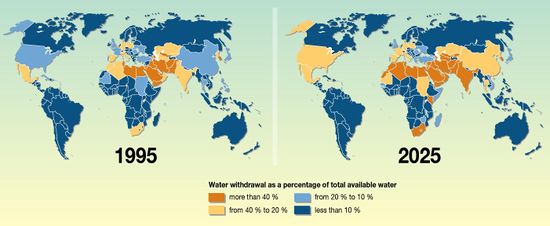
The Durban area in South Africa has been selected as the location of this project since it is currently experiencing approximately 25% water withdrawal as a percentage of total available water. This value is projected to increase in the upcoming years to 40% water withdrawal due to physical water scarcity [3,4] as can been seen in Figure 1. | |||
[[File:MapABC.jpg | thumb | center | 550 px | alt=Alt text| Fig 1: Comparison of water scarcity between 1995 and 2025.]] | |||
In addition to the increasing water scarcity in the next decade, this location was selected due to relatively low location costs and salinity levels as seen in Appendix A. South Africa has a location cost factor of 1.1, which is only 10 % more than the Gulf Coast basis. The country also has relatively low cost of labor and has a labor productivity factor of 1.3 [5]. To provide energy for this process, hydroelectric power from a plant located approximately 200 km away from the proposed location will be used, allowing for a more sustainable energy source at a stable price point. Five desalination plants already exist in the country; all located on the western coast, in suburban areas as shown in Appendix B. A plant located on the eastern side, specifically in Amanzimtoti, near the large metropolis of Durban, would supplement these existing facilities by increasing the amount of available drinking water and provide an economic boost to an area that has one of the largest percentages of agricultural households in the country. | |||
==Preliminary Market Analysis and Process Selection== | ==Preliminary Market Analysis and Process Selection== | ||
There are currently five large scale water desalination plants in South Africa, run by Veolia Water Technologies, as shown in Appendix B [6]. Similar to 80% of desalination plants in the world, South Africa largely utilizes reverse osmosis technology. The 20% of facilities that utilize thermal desalination account for 50% of all water desalinated, but these plants are largely concentrated in the Gulf States due to the high salt concentration of the sea water present and the abundance of cheap fuel. Reverse osmosis technology continues to improve, increase in efficiency, and recently gained market popularity as a desalination technology [2]. Furthermore, the salt concentration in South Africa is average, so multi-stage flash distillation is not required to reach the required drinking water purity. Therefore, reverse osmosis has been selected for this project because at lower inlet salt concentrations, it is more economically viable. | |||
==Process Feed and Composition== | ==Process Feed and Composition== | ||
The salinity of the ocean water around the coast of South Africa is approximately 35 ppt, at 35 g of salt per every 1000 g of water [7]. The seawater inlet of the proposed desalination plant will have the chemical composition shown in Appendix C [8]. In addition to inorganic species in seawater, the inlet will also include heterotrophic bacteria at a concentration of 2.01*109 cells/L [9] and viruses at a concentration of 50*109 viruses/L [10]. Typical limits of total dissolved solids (TDS) are at 1 g/L for drinking water [7]. The products of this desalination process will be lower than this limit, at approximately 0.75 g/L. Individual mineral components must be lower than the upper limits shown in Appendix C to prevent negative health effects [11]. Based on the population of the greater Durban area, the total water intake of the city is estimated to be 2.9 million m3 of water per day [8]. To satisfy the drinking needs of 3.5% of this population, which is the typical output of a desalination plant, the goal of this process is to produce at least 100,000 m3 of freshwater per day. | |||
=Process Alternatives= | =Process Alternatives= | ||
==Pretreatment== | ==Pretreatment== | ||
Pretreating seawater is important to prevent particle, colloidal, organic, mineral, oxidant, and biological fouling. Particle fouling is caused by sand, clay, and suspended solids in seawater. This can be avoided by using filtration. Colloidal and organic fouling can be avoided by using a combination of coagulation and filtration, although the preferred method is ultrafiltration. To remove any final particulates, nanofiltration (5-micron) is required before the feed water enters the RO membranes [12]. A summary of these fouling types and methods to prevent them can be seen in Appendix D. | |||
Biological fouling is caused by bacteria, viruses, microorganisms, and protozoa, which contribute to biofilm formation on the membrane surface. Chlorination during pretreatment kills these microorganisms to prevent biofouling on the membranes. Existing plants typically use sodium hypochlorite injections of 3 mg/L to achieve this. Other options for chlorination include the use of chlorine gas or chlorine dioxide, but sodium hypochlorite is preferred at this stage because it has the lowest operating cost. After killing bacteria, it is necessary to dechlorinate the water because chlorine oxidizes the membranes in the RO process. One possible method for dechlorination is the injection of sodium bisulfate, which results in harmful byproducts, including hydrochloric acid. To prevent this, dechlorination can be achieved using an activated carbon filter, which has been selected for this process. These filters have a very high probability of adsorbing chlorine, making them the optimal choice for dechlorination [12]. | |||
==Membrane Selection== | ==Membrane Selection== | ||
Water treatment processes use several types of membranes, including microfiltration, ultrafiltration, nanofiltration, and reverse osmosis. Microfiltration membranes have the largest pores, followed by ultrafiltration, nanofiltration and finally reverse osmosis [13]. RO membranes filter out monovalent and multivalent ions, viruses, bacteria, and suspended solids [14]. This makes RO membranes the ideal choice for the process, especially if the water is to be consumed. | |||
RO membranes are either made of cellulose acetate or polysulfone coated with aromatic polyamides [13]. Cellulose-based membranes have NaCl rejection values of up to 99.5% at 2000 psig and are often made from acetylated cellulose. Thin Film Composite (TFC) membranes are based on aromatic polyamides, and their characteristics surpass those of acetylated cellulose membranes. They have an average rejection of 99.5% at only 225 psig of feed pressure, making the process more economical and energy efficient. The membranes are made from a highly permeable support of polysulfone and are coated with a cross-linked aromatic polyamide thin film, which allows for the high salt rejection percentage [13]. | |||
There are four types of modules for membrane arrangements: plate-and-frame, tubular, spiral wound, and hollow fiber. An illustration of these types can be seen in Appendix E. The plate-and-frame module is the most simple, containing two end plates, a flat membrane, and spacers. The tubular module is an annulus with the membrane lining the walls and solution flowing through the tube. The spiral wound option has a flat membrane in the form of a sheet that is wrapped around a perforated collection tube. Finally, hollow fiber modules are bundles of fibers in a pressure vessel [13]. | |||
TFC membranes in the spiral wound arrangement have been chosen for this process. TFC membranes reject small organics that could pass through cellulose-based membranes, withstand a larger pH range and can be used at higher temperatures. The spiral wound arrangement was chosen because it has a large surface area and is easier to clean. Hollow fiber membranes provide a higher surface area, but they also incur high maintenance and replacement costs. | |||
==Energy Recovery== | ==Energy Recovery== | ||
There are two main methods for energy recovery: direct recovery of mechanical energy, and conversion to electricity. Direct recovery of the mechanical energy from the high pressure concentrate is the most common method of energy recovery [9]. The devices used to accomplish this can have up to a 98% efficiency. With this type of energy recovery, the high pressure from the concentrate can only be used to pressurize another stream of water. Alternatively, conversion of energy of the high pressure concentrate into electrical energy is much more versatile, but much less efficient. While energy capture from water can be as high as 95% as in hydroelectric power plants, the devices using that energy will likely have a much lower efficiency [10]. For example, a typical pump has an efficiency from 40% to 60%, resulting in much higher overall energy loss. Therefore, for the proposed design, since versatility is not as big of a concern as energy conservation, a direct recovery system will be used for the mechanical energy from the high pressure concentrate to pressurize the filtered water for reverse osmosis. | |||
==Remineralization== | ==Remineralization== | ||
Reverse osmosis membranes cannot selectively remove certain species. Since typical drinking water contains some salts, it is necessary to reintroduce them before water distribution and use. There are four major methods used for water remineralization after reverse osmosis. These methods utilize a mixture of three smaller processes: mixing with saltwater, mixing with salts or gases, and percolation with salt-based filters. Typically, percolation yields the best water quality, but also requires the largest investment and has the highest operating costs [15]. Method overviews are given in Appendix F. All of the methods commonly used bring the water to an acceptable range for drinking water in South Africa. For the proposed design, the process of mixing the reverse osmosis product with brine will be used. While this slightly produces lower water quality than its alternatives, the process still meets all federal guidelines for drinking water and chlorine concentration will be kept below 100 mg/L to prevent corrosion. | |||
==Disinfection== | ==Disinfection== | ||
Although the product stream should not contain any bacterial or viral materials due to the effectiveness of the filtration and RO steps, disinfection is typically done as a precaution. This ensures that water quality is maintained despite any upstream issues with membrane integrity. Chlorine gas, sodium hypochlorite, ozone, and UV light are options for disinfection [16]. UV treatment and ozone are environmentally friendly and have no chemical byproducts, but the costs tend to be much higher. Chlorine gas and sodium hypochlorite have low capital investment and operating costs, but the byproducts can be difficult to manage and are not environmentally safe. Chlorine dioxide treatment is highly effective, requires very low capital investment, and produces byproducts that are easier to manage. While the operating costs are slightly higher than that of chlorine gas and sodium hypochlorite treatment, chlorine dioxide treatment has the best combination of environmental friendliness, overall investment, and effectivity [16]. | |||
==Waste Treatment== | ==Waste Treatment== | ||
An essential factor of the proposed reverse osmosis plant is waste treatment. Outlet streams of desalination plants include hyper-saline solutions at densities higher than circulating ocean water. This difference in density will cause the brine to settle to the ocean floor, potentially resulting in negative effects on the fauna and flora [17]. Conventional brine disposal methods include surface water and sewer discharge, deep well injection, and evaporation ponds [18]. | |||
Surface water discharge directly disposes hyper-saline solutions to surface water. This method has low capital, operating, and manufacturing costs. Although South Africa has less stringent policies on waste eliminations into surface water, ethical considerations should still be taken into account to eliminate negative environmental effects. Sewer discharge directly disposes brine outlet solutions into the sanitary sewer system. This method is cost effective when there are sewer systems nearby the plant. However, this is not the case for our location [18]. Deep well injection disposes brine into porous subsurface rock formations. While this method may be more environmentally friendly, it requires extremely high capital costs. Site evaluation would require time and money, and an extensive scale up analysis would be required to ensure that this method would be feasible. Evaporation ponds utilize solar energy to reduce the water content in brine solutions. This method incurs low costs and is reliable, as few pieces of equipment are required. However, depending on the amount of brine removed from the plant, large amounts of land may be needed to contain the effluents. | |||
The most common technique for brine disposal used by water desalination plants is to discharge back into the ocean [19]. This is legal in South Africa as evidenced by the largest desalination plant in the country at Mossel Bay [20]. According to regulations, a permit must be acquired to discharge large amounts of waste into the ocean [21], but the precedent from Mossel Bay demonstrates that South Africa will grant such a permit to a water desalination plant. In addition, research of nearby protected marine environments was conducted, and it was concluded that the proposed plant location near Amanzimtoti is at least 25 km from the nearest Marine Protected Area [22]. Therefore, direct brine disposal is the chosen alternative for the process. | |||
=Process Overview= | =Process Overview= | ||
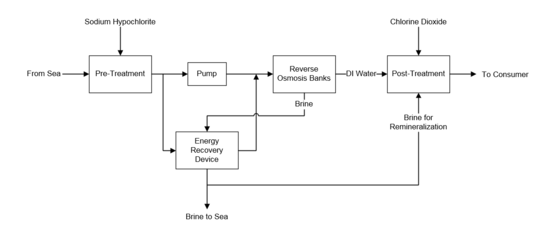
Raw seawater is taken from the ocean and pumped to the pretreatment facility. Sodium hypochlorite is added to remove particulates and biological organisms that would cause fouling in the RO membranes. Pretreatment involves a series of filters to remove the majority of the particulates [13] and an activated carbon filter to dechlorinate the pretreated water before it is pumped to high pressure. Reverse osmosis occurs in two banks of spiral wound membrane modules. The high pressure concentrate exits the reverse osmosis system and is sent to an energy recovery device. This converts mechanical energy from the pressurized concentrate into mechanical energy that pumps the low pressure filtered seawater. The low pressure concentrate is then returned to the ocean. The purified product from reverse osmosis is sent to posttreatment where it is remineralized by mixing with a slight percentage of pressurized concentrate in order to restore the necessary minerals and to meet drinking water standards. It is then disinfected with chlorine dioxide and sent to the consumer. See Figure 2 for a block flow diagram of this process. | |||
[[File: Figure2RHP.png | thumb | center | 550px | alt=Alt text | Fig 2: Block flow diagram of the proposed desalination process.]] | |||
=Process Flow Diagram= | |||
See Appendix G for the full process flow diagram. The process has been divided into two sections to aid in organization. The pretreatment section is represented by the 100 series of equipment while the reverse osmosis and posttreatment section is represented by the 200 series. A detailed layout of the reverse osmosis modules is provided in Figure G3 in Appendix G. | |||
In the pretreatment step, a cascade of filters is implemented in the following order: macrofiltration, microfiltration, activated carbon, ultrafiltration and nanofiltration. Three pumps are utilized to account for the pressure drops across these filters. After pretreatment, the clarified seawater stream is split and sent to a pump and an energy recovery device to pressurize the reverse osmosis feed. After going through both reverse osmosis banks, the deionized water is remineralized with brine and disinfected with chlorine dioxide. The brine is used in the energy recovery device, then returned to the sea. | |||
Control systems were added to this process to maintain safe operations and a standard level of product quality. For pretreatment, a fail-close system measures the level of seawater in V-100 and regulates the amount of sodium hypochlorite needed to disinfect the seawater. In the 200 series, two control loops were implemented, including a fail-close pressure control loop that maintains the necessary pressure by monitoring the concentrate entering C-200. A fail-open level controller measures the amount of purified water stored before post-treatment. The valve can increase the inlet flow rate of the final storage tank if the liquid level of purified water exceeds the set-point. This by no means encompasses all the control that should be implemented in the process and only serves as a representation of the necessity of these systems. | |||
=Mass and Energy Balances= | |||
The feed stream is assumed to be at the average seawater temperature of around 25℃ and at 1 atm [23]. The inlet water stream is estimated to have 500 ug/L of particulate matter. Flow rates, conditions, and compositions of each stream can be found in the stream table in Appendix H. | |||
Pressure drops calculated for each pump and filtration step to estimate the utility requirements in the process can be found in Appendix I. The Ergun equation was used to calculate the pressure drops across each level of macrofiltration, which removes all particulate matter. Imperfect packing was assumed, with a void fraction of 0.4. The particle diameter was based off of the Wentworth Grain Size Chart [24], which can be found in Appendix J. The diameter of the filtration tank is 3.5 m because having a larger cross-sectional area decreases velocity. The pressure drops for the microfiltration, ultrafiltration, and nanofiltration modules were calculated using the Darcy equation and transmembrane pressure relationships from literature [25]. | |||
Granular activated carbon (GAC) filtration will be used for the dechlorination of the pretreated water. According to the US EPA, surface loading rates for GAC filters range between 2 to 10 gpm per square foot [26]. Using this typical flow rate, the pressure drop of flow through the activated carbon filter was estimated to be 0.6 psi per foot of bed depth found in Appendix K [27]. The activated carbon filter is assumed to effectively remove all of the hypochlorite from the stream. | |||
The amount of brine used to remineralize the permeate ensures that the chlorine levels of the drinking water outlet remain below the upper limit set by South African regulations, as stated in Appendix C. The required concentrations of active chlorine in pretreatment and posttreatment to effectively disinfect the streams are 3 mg/L and 0.2 mg/L, respectively [28, 29]. | |||
=Design Constraints= | |||
Sustainability has been a strong motivator throughout the design process. An energy recycle system has been implemented to combat this issue. This allows for cheaper production cost and lower energy requirements. Further sustainability efforts could include utilizing renewable energy sources such as solar, wind, and nuclear power. Environmental and ethical considerations are involved with releasing brine back into the sea. Although there are no laws controlling the release of concentrate back into the ocean, the process has been designed to carefully consider the impact and location of the release. The process avoids dangerous effects of this action by being located in an area far from declared marine protected areas. In addition, the brine will be released 500 m away from shore, decreasing its impact on coastal marine life. | |||
The most pressing ethical consideration is the required level of water quality. By selecting a location outside of the United States, different regulations and standards could be applied to the purified drinking water that will be sent to consumers. The drinking water produced in this process will not only meet the drinking water standards of South Africa but will also exceed those limits to provide a healthier and cleaner water alternative allowing for a better drinking water experience. | |||
Throughout the plant, safety considerations heavily impact the specifics of the process. Multiple control loops were implemented to ensure effective and safe production. The chloride concentration is near its maximum possible level. If the chloride ion concentration increases, it could cause increased corrosion. Therefore, to ensure a longer lifetime for the process, all pipes in the system are to be made with polyethylene. An increase in the corrosiveness of the water could cause metals ions to contaminate the water with certain types of piping as is currently being seen in Flint, Michigan. Therefore, chloride ion concentration will be carefully monitored to prevent negative health effects. | |||
=Equipment Sizing and Trade-offs= | |||
Equipment was sized to ensure that capital costs are not unreasonably high while satisfying the design basis requirements. The macrofiltration tank is filled with large amounts of sand with different levels of coarsity. The tank was designed to be as wide as possible at 3.5 meters to minimize the pressure drop and pumping requirements. For the amount of water being filtered daily, roughly 50 kg of particulate matter will accumulate. At an approximate density of 3,000 kg/m3, this would result in an accumulation of 16.7 liters of particulate matter per day. Thus, with a total height of 2 meters, there should be enough space to accommodate both sand and accumulated particulate matter. | |||
Filters are sold as compact modules containing a large surface area. Increasing the area of the filters decreases the pumping requirements of the system, but increases the capital cost of purchasing the required filters. For each filtration step, many of these units will be used in parallel to meet the filtration surface area requirements, shown in Appendix L. Care was also taken to ensure that the pressure drop across the filters did not exceed product specifications. | |||
The system incorporates three storage tanks. The first tank has a inlet flow rate of 7080 m3/hr, and the second and third tanks each have inlet flows of 5670 m3/hr. Because these flow rates are high, the tanks cannot hold water for long periods of time. The storage capacity for this facility was chosen to be 1.5 hours’ worth of flow. With two parallel units at each step to ensure repairs can be made efficiently, the total storage capacity of this facility will be about 64,000 m3 [30]. | |||
Dead end filters were chosen for all of the filtration steps. This increases the capital cost required as the cleaning process for dead end filtration necessitates the purchase of an excess of membranes. Filters will be active for 60 seconds and subsequently cleaned for 5 seconds while the flow is rerouted to another set of membranes. During cleaning, air is sent through the filters in the direction opposite the flow. Purchasing an excess of filters ensures that the required surface area is always available even though roughly 1/13th of all filters will be undergoing cleaning at any moment [31]. The benefit of dead end filtration is that it results in a higher yield, and therefore lower pumping requirements overall. | |||
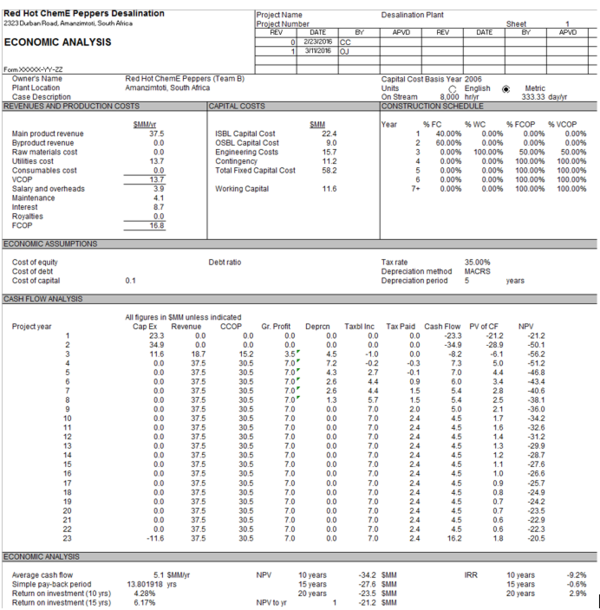
=Economic Analysis= | |||
The current capital cost for the project is $22.4 MM, with an OSBL capital cost of $8.96 MM, as predicted by the Aspen Process Economic Analyzer. This capital cost includes the equipment cost, $18.4 MM, the membrane costs for pretreatment and reverse osmosis, $2.1 MM, and piping, land, and facility costs totaling $1.9 MM. Detailed breakdown of equipment and membrane costing can be seen in Appendix M. Equipment costs have been estimated using fiberglass reinforced plastic for construction of tanks and using polyethylene for piping to reduce corrosion [32]. Periodic backwashing is required to remove any particulates that gather during filtration; this cost has been accounted for in maintenance, which was estimated to be 7% of the ISBL investment [33, 34]. | |||
The main revenue of producing approximately 132,000 m3 per day of desalinated water sold at $0.81 per metric ton is $37.5 MM [35]. Utility costs for this design are $13.7 MM per year. This value arises from the requirement of 20,209 KW of electricity purchased at $0.08 per KWh [36]. To decrease the environmental impact from the large utility requirement, electricity will be obtained from the Ingula Pumped Storage Scheme [37], a hydropower plant located approximately 250 km away from our proposed location. | |||
Raw material costs for this project are relatively low, approximately $0.04 MM. This is mainly due to the fact that the seawater is obtained less than 500 m away from the coast and does not require transport or processing. The sodium hypochlorite for chlorination during pre-treatment can be purchased at $108 per metric ton [38], and the chlorine dioxide can be purchased at approximately $2200 per metric ton [39]. | |||
Land cost was estimated using online property listings by finding different vacant lots near Amanzimtoti. The cost per acre was estimated to be R550,000 or $35,000. The required land area is approximately 8 acres [40], resulting in total land costs of approximately $300,000. The predicted salary and overheads are $0.8 MM, which include 3 shifts of 5 operators at a salary of R150,000 or $9,500 per operator, the average salary for a plant operator in South Africa [41]. | |||
Using typical industrial economic relationships, the OSBL was estimated to be 40% of the ISBL, the engineering costs to be 30% ISBL plus OSBL, the contingency charge to be 10% of the ISBL plus OSBL, and working capital to be 20% of the total fixed capital cost. The construction schedule models plant building during the first two years at 40% and 60% investment, respectively. Full production is set to begin immediately upon plant completion. The depreciation rate chosen was the 5 year MACRS method because it yields better return than the straight-line model. The tax rate was selected to be 35%, reflecting the standard industrial tax rate in South Africa [42]. | |||
The net present value at 10 years is predicted to be -$34.2 MM and increases to -$23.5 MM at 20 years with an internal return rate is 2.9% at 20 years. As of now, the process is not economically feasible. However, as described by the sensitivity analysis below, the process may become economically viable if utility costs and product revenue change favorably. | |||
=Optimization and Sensitivity Analysis= | |||
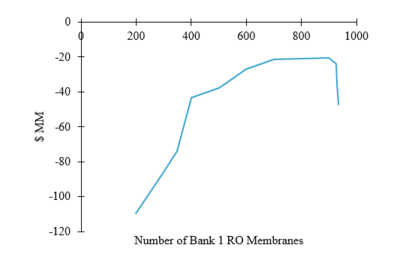
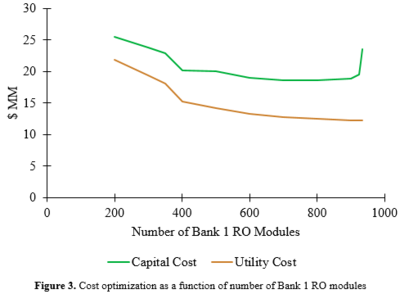
A cost optimization was performed to determine how reverse osmosis modules could be organized to minimize costs. As the number of modules increases, the capital and operating costs decrease as seen in Figure 3. The module costs are trivial in comparison to the capital costs of the pumps. As the number of modules approaches 900, utility requirements approach a minimum value, but the number of modules needed in the second bank begins to increase exponentially, causing a spike in the capital costs. As a result, the optimal balance between operating costs and utility costs was found to be 900 modules in the first bank and 4000 modules in the second bank. | |||
[[File:Figure3RHP.PNG |thumb|center|400px|alt=Alt text|Fig 3: Cost optimization as a function of number of Bank 1 RO modules]] | |||
A sensitivity analysis was performed to elucidate the effect of four major variables on the NPV at the end of 20 years: selling price of water, maintenance cost, operating cost, and capital cost. Each variable was modified between 75% and 125% of its present value to determine its effect on the NPV. Overall, the price of water has the largest impact, followed by the operating costs, capital costs, and the maintenance costs, as seen in Figure 4. | |||
[[File:Figure4RHP.PNG |thumb|center|400px|alt=Alt text|Fig 4: Sensitivity analysis showing changes in NPV as prices and costs change]] | |||
Although the NPV is currently negative, the sensitivity analysis shows a path towards having a positive NPV. If the price of water increases by 15% and the cost of energy decreases by 15%, the NPV reaches a positive value. These are values that could potentially be reached through negotiations with various government agencies and energy providers. It may be possible to obtain electricity at a lower price due to the construction of a 3,000 MW hydroelectric dam in 2017. Additionally, due to the rising water scarcity in Africa, an increase in the price of water can be expected in the future. | |||
=Conclusion= | |||
The proposed process meets the design requirements of producing 100,000 m3 per day of clean drinking water. However, due to high operating costs and significant capital costs, the design is not economically viable, due to a 20 year NPV of -$23.5 MM. A sensitivity analysis showed that the price of water is most important for determining profitability. Currently, the selling price of water is too low for the plant to be profitable. If the plant were to be constructed, contracts could be negotiated between suppliers and electricity providers in order to provide a consistent lower price. Therefore, it is not recommended to implement the plant design unless there are significant changes in the market for drinking water in South Africa. | |||
=References= | |||
#International Decade for Action 'Water for Life'. UN News Center. Available at: http://www.un.org/waterforlifedecade/scarcity.shtml. Accessed January 13, 2016. This source was used to find statistics about water scarcity to better understand the problem. | |||
#Greenlee LF, Lawler DF, Freeman BD, Marrot B, Moulin P. Reverse osmosis desalination: water sources, technology, and today's challenges. Water Res. 2009;43(9):2317-48. This source contains specifics about the reverse osmosis process and similar technologies. | |||
#United Nations Environment Programme. Increased global water stress. 2008. http://www.unep.org/dewa/vitalwater/article141.html | |||
#Madison Powers. Water Scarcity. 2015. http://www.fewresources.org/water-scarcity-issues-were-running-out-of-water.html | |||
#Humphreys, K.K. Sources of International Cost Data. Presented at NORDNET ‘97 Conference. Reykjavik, Iceland. September 11, 1997. | |||
#Water purification plant solutions. Veolia. Available at: http://www.veoliawaterst.co.za/municipal-water-treatment/water-purification/water-purification-plant/. Accessed January 13, 2016. This source was used to determine locations for other desalination plants in South Africa. | |||
#Map of ocean salinity. Science Learning Hub RSS. Available at: http://sciencelearn.org.nz/contexts/the-ocean-in-action/sci-media/images/map-of-ocean-salinity. Accessed January 13, 2016. This source was used to classify the salinity of the water surrounding South Africa. | |||
#IDE's Israel seawater RO desalination plant sets world record for water production. - WaterWorld. Available at: http://www.waterworld.com/articles/2015/02/ide-s-israel-seawater-ro-desalination-plant-sets-new-world-record.html. Accessed January 15, 2016. This source was used to determine the water intake of the greater city of Durban for plant capacity sizing. | |||
#Li WKW. Annual average abundance of heterotrophic bacteria and Synechococcus in surface ocean waters. Limnology and Oceanography 1998;43(7):1746–1753. Available at: http://www.aslo.org/lo/toc/vol_43/issue_7/1746.pdf. Accessed January 15, 2016. This source was used to determine the concentration of bacteria in seawater. | |||
#Pearson - Science News. Pearson - Science News. Available at: http://www.phschool.com/science/science_news/articles/all_worlds_phage.html. Accessed January 15, 2016. This source was used to determine the concentration of viruses in seawater. | |||
#Holmes S. South African Water Quality Guidelines. 2nd ed. Pretoria: The Dept. of Water and Forestry; 1996. | |||
#Water Treatment Solutions. (n.d.). http://www.lenntech.com/processes/desalination/pretreatment/general/desalination-pretreatment.htm Accessed January 22, 2016 This source was used to determine pretreatment for RO. | |||
#Sagle A, Freeman B. Fundamentals of Membranes for Water Treatment. The Future of Desalination in Texas. 2004;2. http://www.twdb.texas.gov/publications/reports/numbered_reports/doc/r363/c6.pdf Accessed January 28, 2016. This source provides information about different types of membranes for RO filtration. | |||
#Membrane Technologies. Koch Membrane Systems. http://www.kochmembrane.com/Learning-Center/Technologies.aspx Accessed January 28, 2016. This source provides the effectiveness of RO membranes. | |||
#Desalination Post-treatment: Neutralization/Remineralization for Drinking Water. Lenntech. http://www.lenntech.com/processes/desalination/post-treatment/post-treatments/remineralization.htm Accessed January 20, 2016. This source provides a description of different remineralization systems. | |||
#Disinfection. Lenntech. http://www.lenntech.com/processes/disinfection/disinfection.htm Accessed January 29, 2016. This source provides options for disinfection. | |||
#Ocean Standards. California Environmental Protection Agency: State Water Resources Control Board. http://www.waterboards.ca.gov/water_issues/programs/ocean/desalination/ Accessed January 20, 2016. This source provides the impact of brine on the environment. | |||
#Brandhuber P, Cerone J, Kwan P, Moore E, Vieira A. A look at conventional and emerging brine disposal and waste minimization technologies. HDR Waterscapes. 2008; 9; 2. https://www.hdrinc.com/sites/all/files/content/articles/article-files/3601-a-look-at-conventional-and-emerging-brine-disposal-and-waste-minimization-technologies.pdf Accessed January 20, 2016. This source provides an overview of the alternatives available for brine disposal. | |||
#Tularam GA, Ilahee M. Environmental concerns of desalinating seawater using reverse osmosis. Journal of Environmental Monitoring. 2007; 9; 805-813. http://pubs.rsc.org/en/content/articlehtml/2007/EM/B708455M Accessed January 27, 2016. This source states that discharging back into the ocean is the most common method of disposal. | |||
#South Africa’s Largest Seawater Desalination Plant. Veolia Water Solutions & Technologies. http://www.veoliawaterst.co.za/vwst-southafrica/ressources/files/1/32048,Mossel-Bay-Desalination.pdf Accessed January 27, 2016. This source provides information on the desalination plant at Mossel Bay. | |||
#Department of Environmental Affairs. National Guideline for the Discharge of Effluent from Land-based Sources into the Coastal Environment. 2014. https://www.environment.gov.za/sites/default/files/legislations/nationalguideline_landbasedinfluent_dischargecoastal.pdf Accessed January 27, 2016. This source provides information on obtaining a permit for discharging effluent back into the ocean. | |||
#Tunley K, Chadwick P, Nobula S. State of Management of South Africa’s Marine Protected Areas. WWF South Africa Report Series. 2009. http://assets.wwfza.panda.org/downloads/mpastateofmanagementreport04nov2009weblowerdpi.pdf Accessed January 27, 2016. This source provides locations of marine protected areas near South Africa. | |||
#Global Sea Temperatures - Durban Average Monthly Sea Temperature. Sea Temperature. http://www.seatemperature.org/africa/south-africa/durban-february.htm Published 2016. Accessed February 18, 2016. | |||
#U.S. Geological Survey. Surficial Geology of the Sea Floor in Long Island Sound Offshore of Orient Point, New York. USGS. http://pubs.usgs.gov/of/2010/1100/html/fig10.html Accessed February 18, 2016. | |||
#Crittenden J. Water Treatment: Principles and Design. Wiley; 2005. | |||
#Drinking Water Treatability Database. Granular Activated Carbon. U.S. Environmental Protection Agency. http://iaspub.epa.gov/tdb/pages/treatment/treatmentOverview.do?treatmentProcessId=2074826383 Published 2016. Accessed February 18, 2016. | |||
#Clack Corporation. Granular Activated Carbon (GAC). http://www.farriswater.com/pdf/gac.pdf Published August 13, 2008. Accessed February 18, 2016. | |||
#Lenntech. Chemical Disinfectants - Chlorine. http://www.lenntech.com/processes/disinfection/chemical/disinfectants-chlorine.htm Accessed February 19, 2016. | |||
#Lenntech. Chemical Disinfectants - Chlorine Dioxide. http://www.lenntech.com/processes/disinfection/chemical/disinfectants-chlorine.htm Accessed February 19, 2016. | |||
#Alibaba.com. Building fiberglass cooling water storage tank. http://www.alibaba.com/product-detail/building-fiberglass-cooling-water-storage-tank_60019235269.html?spm=a2700.7724838.30.119.kzLzuC Accessed Feb 27, 2016. | |||
#Lenntech. Membrane Cleaning Methods. http://www.lenntech.com/membrane-cleaning.htm Accessed Feb 29, 2016. | |||
#Alibaba.com. Underground water large diameter plastic drip irrigation pipe. http://www.alibaba.com/product-detail/underground-water-large-diameter-plastic-drip_60213689907.html?spm=a2700.7724838.0.0.NrgLIM Accessed Feb 29, 2016. | |||
#DeSilva, F. Water Treatment Guide. Activated Carbon Filtration. http://www.watertreatmentguide.com/activated_carbon_filtration.htm Published January 2000. Accessed Feb 29, 2016. | |||
#Muilenberg, T. Water and Wastes Digest. Microfiltration: How does it compare? http://www.wwdmag.com/desalination/microfiltration-how-does-it-compare Published December 28, 2000. Accessed Feb 29, 2016. | |||
#Sweet, P. Las Vegas Sun. Desalination Gets a Serious Look. http://lasvegassun.com/news/2008/mar/21/desalination-gets-serious-look/ Published March 28, 2008. Accessed Feb 29, 2016. | |||
#Business Tech. Electricity Prices: South Africa vs the World. http://businesstech.co.za/news/energy/99494/electricity-prices-south-africa-vs-the-world/ Published September 2015. Accessed Feb 28, 2016. | |||
#Eskom. Ingula Pumped Storage Scheme. http://www.eskom.co.za/Whatweredoing/NewBuild/IngulaPumpedStorage/Pages/Ingula_Pumped_Storage_Scheme.aspx Accessed Feb 27, 2016. | |||
#USP Technologies. Sodium Hypochlorite. http://www.h2o2.com/products-and-services/us-peroxide-technologies.aspx?pid=116 Published 2016. Accessed Feb 29, 2016. | |||
#www.proformindustries.com/~proform/sites/.../purewater_presentation.pptx | |||
#Tampa Bay Water. Seawater Desalination Plant Data Sheet. http://www.tampabaywater.org/documents/fact-sheets/desal-fact-sheet.pdf Published 2010. Accessed Feb 27, 2016. | |||
#PayScale Human Capital. Average Salary for People with Jobs as Plant Operators/Managers/Supervisors. http://www.payscale.com/research/ZA/People_with_Jobs_as_Plant_Operators%2FManagers%2FSupervisors/Salary Accessed Feb 29, 2016. | |||
#Trading Economics. South Africa Corporate Tax Rate. http://www.tradingeconomics.com/south-africa/corporate-tax-rate Accessed Feb 27, 2016. | |||
=Appendices= | |||
==Appendix A== | |||
This appendix includes a map representing the salinity of seawater around the world in parts per thousand. | |||
[[File:FigureA1RHP.png|thumb|center|400px|alt=Alt text|Fig A1: A representation of salinity levels around the world.]] | |||
==Appendix B== | |||
This appendix includes the locations of the five existing desalination plants in South Africa. | |||
[[File:FigureB1RHP.png|thumb|center|400px|alt=Alt text|Fig B1: A map of existing locations of desalination plants in South Africa]] | |||
#Knysa - Western Cape | |||
#Plettenberg Bay | |||
#Mossel Bay | |||
#Lamberts Bay | |||
#Cannon Rocks and Boknes Communities | |||
==Appendix C== | |||
This appendix includes the inorganic compositions in seawater along the coast of South Africa, as well as the upper limits for each of these species. | |||
[[File:TableC1RHP.PNG|thumb|center|400px]] | |||
==Appendix D== | |||
This appendix provides summaries of process alternatives for pretreatment. | |||
[[File:TableD1RHP.PNG|thumb|center|400px]] | |||
==Appendix E== | |||
This appendix includes the different types of reverse osmosis membrane modules. | |||
[[File:FigureE1RHP.png|thumb|center|400px|alt=Alt text|Fig E1: Four types of RO membrane modules: a) plate and frame, b) tubular, c) spiral wound, and d) hollow fiber]] | |||
==Appendix F== | |||
This appendix includes the different alternatives for remineralization and the included comparison of capital and operating costs. | |||
[[File:TableF1RHP.PNG|thumb|center|400px]] | |||
==Appendix G== | |||
This appendix contains the process flow diagrams divided into the 100 section for pretreatment and the 200 section for reverse osmosis and posttreatment. A layout of the banks for reverse osmosis is also included. | |||
[[File:FigureG1RHP.png|thumb|center|600px|alt=Alt text|Fig G1: Process flow diagram of pretreatment]] | |||
[[File:FigureG2RHP.png|thumb|center|600px|alt=Alt text|Fig G2: The process flow diagram of reverse osmosis and posttreatment]] | |||
[[File:FigureG3RHP.png|thumb|center|600px|alt=Alt text|Fig G3: Detailed process flow diagram of reverse osmosis membranes]] | |||
==Appendix H== | |||
This appendix includes stream conditions and composition in the process. | |||
[[File:TableH1RHP.png|thumb|center|400px]] | |||
[[File:TableH2RHP.png|thumb|center|400px]] | |||
==Appendix I== | |||
This appendix includes the energy requirements for each pump in the process. | |||
[[File:TableI1RHP.PNG|thumb|center|400px]] | |||
==Appendix J== | |||
This appendix includes the Wentworth Grain Size Chart used for pressure drop calculations of the macrofiltration step in pretreatment. | |||
[[File:FigureJ1RHP.PNG|thumb|center|400px]] | |||
== | ==Appendix K== | ||
This appendix includes the calculated pressure drops across all the membranes used in the process. | |||
[[File:TableK1RHP.PNG|thumb|center|350px]] | |||
== | ==Appendix L== | ||
This appendix includes the results of the surface area calculations for each step in the process. | |||
[[File:TableL1RHP.PNG|thumb|center|400px]] | |||
== | ==Appendix M== | ||
This appendix includes the economic analysis spreadsheet, along with the costs of production and a graph showing the resulting net present value with changing the number of reverse osmosis membranes in the first bank of the system. | |||
[[File:FigureM1RHP.PNG|thumb|center|600px|alt=Alt text|Fig M1: Cost of Production]] | |||
[[File:FigureM2RHP.PNG|thumb|center|600px|alt=Alt text|Fig M2: Economic Analysis]] | |||
[[File:FigureM3RHP.PNG|thumb|center|400px|alt=Alt text|Fig M3: Optimization of net present value by number of reverse osmosis membranes in first bank]] | |||
Latest revision as of 23:13, 11 March 2016
Reverse Osmosis Desalination Plant in Durban, South Africa
Authors: Lauren Burke, Cindy Chen, Osman Jamil, Natalia Majewska
Instructors: Fengqi You, David Wegerer
March 11, 2016
Executive Summary
An imminent global shortage of water will greatly affect the lives of millions of people. The situation in South Africa is predicted to exacerbate in the upcoming decades. This growing need for fresh drinking water motivated the design of a reverse osmosis desalination plant in the suburbs of Durban on the eastern coast of South Africa. A reverse osmosis process was selected based on the water salinity in the area and recent technological advancements in membrane efficiency.
The process is designed to operate for 350 days a year, 24 hours a day to yield 132,000 m3 of water per day. Prior to reverse osmosis, the seawater is pretreated to prevent membrane fouling using a cascade of filters and high-pressure pumps. Reverse osmosis is carried out at 37 bar with two banks containing 900 and 4000 membranes, respectively. After reverse osmosis, the pressurized concentrate that contains the unwanted salts and ions is sent to an energy recovery device to pressurize the reverse osmosis feed. The deionized water from the reverse osmosis system is remineralized and disinfected before being sent to the consumer.
Based on a thorough economic analysis, it has been determined that the process is not feasible at this stage. The annual estimated revenue is $37.5 MM. In addition, the 20 year NPV of the plant is -$23.5 MM with an IRR of 2.9%. A sensitivity analysis indicated that an increase in the price of water could potentially increase profits. Therefore, the design may become profitable implemented with the aid of government subsidies or if a drastic increase in water scarcity increases the selling price of water.
Introduction
Background
Close to 1.2 billion people, approximately one fifth of the world’s population, reside in areas of physical water scarcity [1]. Although there is enough water in the world for a population of seven billion people, it is unevenly distributed, and many regions have been experiencing water shortages for more than a decade. Given the current rate of population growth and water consumption, many studies predict that this problem will be exacerbated in the years to come. Water desalination is a process used to purify water so that it meets drinking standards for consumption. The two main technologies used for desalination are thermal desalination and reverse osmosis. While thermal desalination is a robust method that scales well, it is typically preferable for inlets of very high salt concentrations and very low energy costs, such as the gulf coast region. Reverse osmosis plants are more prevalent elsewhere, making up 80% of all desalination plants in the world [2]. Given the salinity of the inlet flow for this process and advances in technology, reverse osmosis has been selected as the optimal choice for this project.
Location Selection
The Durban area in South Africa has been selected as the location of this project since it is currently experiencing approximately 25% water withdrawal as a percentage of total available water. This value is projected to increase in the upcoming years to 40% water withdrawal due to physical water scarcity [3,4] as can been seen in Figure 1.
In addition to the increasing water scarcity in the next decade, this location was selected due to relatively low location costs and salinity levels as seen in Appendix A. South Africa has a location cost factor of 1.1, which is only 10 % more than the Gulf Coast basis. The country also has relatively low cost of labor and has a labor productivity factor of 1.3 [5]. To provide energy for this process, hydroelectric power from a plant located approximately 200 km away from the proposed location will be used, allowing for a more sustainable energy source at a stable price point. Five desalination plants already exist in the country; all located on the western coast, in suburban areas as shown in Appendix B. A plant located on the eastern side, specifically in Amanzimtoti, near the large metropolis of Durban, would supplement these existing facilities by increasing the amount of available drinking water and provide an economic boost to an area that has one of the largest percentages of agricultural households in the country.
Preliminary Market Analysis and Process Selection
There are currently five large scale water desalination plants in South Africa, run by Veolia Water Technologies, as shown in Appendix B [6]. Similar to 80% of desalination plants in the world, South Africa largely utilizes reverse osmosis technology. The 20% of facilities that utilize thermal desalination account for 50% of all water desalinated, but these plants are largely concentrated in the Gulf States due to the high salt concentration of the sea water present and the abundance of cheap fuel. Reverse osmosis technology continues to improve, increase in efficiency, and recently gained market popularity as a desalination technology [2]. Furthermore, the salt concentration in South Africa is average, so multi-stage flash distillation is not required to reach the required drinking water purity. Therefore, reverse osmosis has been selected for this project because at lower inlet salt concentrations, it is more economically viable.
Process Feed and Composition
The salinity of the ocean water around the coast of South Africa is approximately 35 ppt, at 35 g of salt per every 1000 g of water [7]. The seawater inlet of the proposed desalination plant will have the chemical composition shown in Appendix C [8]. In addition to inorganic species in seawater, the inlet will also include heterotrophic bacteria at a concentration of 2.01*109 cells/L [9] and viruses at a concentration of 50*109 viruses/L [10]. Typical limits of total dissolved solids (TDS) are at 1 g/L for drinking water [7]. The products of this desalination process will be lower than this limit, at approximately 0.75 g/L. Individual mineral components must be lower than the upper limits shown in Appendix C to prevent negative health effects [11]. Based on the population of the greater Durban area, the total water intake of the city is estimated to be 2.9 million m3 of water per day [8]. To satisfy the drinking needs of 3.5% of this population, which is the typical output of a desalination plant, the goal of this process is to produce at least 100,000 m3 of freshwater per day.
Process Alternatives
Pretreatment
Pretreating seawater is important to prevent particle, colloidal, organic, mineral, oxidant, and biological fouling. Particle fouling is caused by sand, clay, and suspended solids in seawater. This can be avoided by using filtration. Colloidal and organic fouling can be avoided by using a combination of coagulation and filtration, although the preferred method is ultrafiltration. To remove any final particulates, nanofiltration (5-micron) is required before the feed water enters the RO membranes [12]. A summary of these fouling types and methods to prevent them can be seen in Appendix D.
Biological fouling is caused by bacteria, viruses, microorganisms, and protozoa, which contribute to biofilm formation on the membrane surface. Chlorination during pretreatment kills these microorganisms to prevent biofouling on the membranes. Existing plants typically use sodium hypochlorite injections of 3 mg/L to achieve this. Other options for chlorination include the use of chlorine gas or chlorine dioxide, but sodium hypochlorite is preferred at this stage because it has the lowest operating cost. After killing bacteria, it is necessary to dechlorinate the water because chlorine oxidizes the membranes in the RO process. One possible method for dechlorination is the injection of sodium bisulfate, which results in harmful byproducts, including hydrochloric acid. To prevent this, dechlorination can be achieved using an activated carbon filter, which has been selected for this process. These filters have a very high probability of adsorbing chlorine, making them the optimal choice for dechlorination [12].
Membrane Selection
Water treatment processes use several types of membranes, including microfiltration, ultrafiltration, nanofiltration, and reverse osmosis. Microfiltration membranes have the largest pores, followed by ultrafiltration, nanofiltration and finally reverse osmosis [13]. RO membranes filter out monovalent and multivalent ions, viruses, bacteria, and suspended solids [14]. This makes RO membranes the ideal choice for the process, especially if the water is to be consumed.
RO membranes are either made of cellulose acetate or polysulfone coated with aromatic polyamides [13]. Cellulose-based membranes have NaCl rejection values of up to 99.5% at 2000 psig and are often made from acetylated cellulose. Thin Film Composite (TFC) membranes are based on aromatic polyamides, and their characteristics surpass those of acetylated cellulose membranes. They have an average rejection of 99.5% at only 225 psig of feed pressure, making the process more economical and energy efficient. The membranes are made from a highly permeable support of polysulfone and are coated with a cross-linked aromatic polyamide thin film, which allows for the high salt rejection percentage [13].
There are four types of modules for membrane arrangements: plate-and-frame, tubular, spiral wound, and hollow fiber. An illustration of these types can be seen in Appendix E. The plate-and-frame module is the most simple, containing two end plates, a flat membrane, and spacers. The tubular module is an annulus with the membrane lining the walls and solution flowing through the tube. The spiral wound option has a flat membrane in the form of a sheet that is wrapped around a perforated collection tube. Finally, hollow fiber modules are bundles of fibers in a pressure vessel [13].
TFC membranes in the spiral wound arrangement have been chosen for this process. TFC membranes reject small organics that could pass through cellulose-based membranes, withstand a larger pH range and can be used at higher temperatures. The spiral wound arrangement was chosen because it has a large surface area and is easier to clean. Hollow fiber membranes provide a higher surface area, but they also incur high maintenance and replacement costs.
Energy Recovery
There are two main methods for energy recovery: direct recovery of mechanical energy, and conversion to electricity. Direct recovery of the mechanical energy from the high pressure concentrate is the most common method of energy recovery [9]. The devices used to accomplish this can have up to a 98% efficiency. With this type of energy recovery, the high pressure from the concentrate can only be used to pressurize another stream of water. Alternatively, conversion of energy of the high pressure concentrate into electrical energy is much more versatile, but much less efficient. While energy capture from water can be as high as 95% as in hydroelectric power plants, the devices using that energy will likely have a much lower efficiency [10]. For example, a typical pump has an efficiency from 40% to 60%, resulting in much higher overall energy loss. Therefore, for the proposed design, since versatility is not as big of a concern as energy conservation, a direct recovery system will be used for the mechanical energy from the high pressure concentrate to pressurize the filtered water for reverse osmosis.
Remineralization
Reverse osmosis membranes cannot selectively remove certain species. Since typical drinking water contains some salts, it is necessary to reintroduce them before water distribution and use. There are four major methods used for water remineralization after reverse osmosis. These methods utilize a mixture of three smaller processes: mixing with saltwater, mixing with salts or gases, and percolation with salt-based filters. Typically, percolation yields the best water quality, but also requires the largest investment and has the highest operating costs [15]. Method overviews are given in Appendix F. All of the methods commonly used bring the water to an acceptable range for drinking water in South Africa. For the proposed design, the process of mixing the reverse osmosis product with brine will be used. While this slightly produces lower water quality than its alternatives, the process still meets all federal guidelines for drinking water and chlorine concentration will be kept below 100 mg/L to prevent corrosion.
Disinfection
Although the product stream should not contain any bacterial or viral materials due to the effectiveness of the filtration and RO steps, disinfection is typically done as a precaution. This ensures that water quality is maintained despite any upstream issues with membrane integrity. Chlorine gas, sodium hypochlorite, ozone, and UV light are options for disinfection [16]. UV treatment and ozone are environmentally friendly and have no chemical byproducts, but the costs tend to be much higher. Chlorine gas and sodium hypochlorite have low capital investment and operating costs, but the byproducts can be difficult to manage and are not environmentally safe. Chlorine dioxide treatment is highly effective, requires very low capital investment, and produces byproducts that are easier to manage. While the operating costs are slightly higher than that of chlorine gas and sodium hypochlorite treatment, chlorine dioxide treatment has the best combination of environmental friendliness, overall investment, and effectivity [16].
Waste Treatment
An essential factor of the proposed reverse osmosis plant is waste treatment. Outlet streams of desalination plants include hyper-saline solutions at densities higher than circulating ocean water. This difference in density will cause the brine to settle to the ocean floor, potentially resulting in negative effects on the fauna and flora [17]. Conventional brine disposal methods include surface water and sewer discharge, deep well injection, and evaporation ponds [18].
Surface water discharge directly disposes hyper-saline solutions to surface water. This method has low capital, operating, and manufacturing costs. Although South Africa has less stringent policies on waste eliminations into surface water, ethical considerations should still be taken into account to eliminate negative environmental effects. Sewer discharge directly disposes brine outlet solutions into the sanitary sewer system. This method is cost effective when there are sewer systems nearby the plant. However, this is not the case for our location [18]. Deep well injection disposes brine into porous subsurface rock formations. While this method may be more environmentally friendly, it requires extremely high capital costs. Site evaluation would require time and money, and an extensive scale up analysis would be required to ensure that this method would be feasible. Evaporation ponds utilize solar energy to reduce the water content in brine solutions. This method incurs low costs and is reliable, as few pieces of equipment are required. However, depending on the amount of brine removed from the plant, large amounts of land may be needed to contain the effluents.
The most common technique for brine disposal used by water desalination plants is to discharge back into the ocean [19]. This is legal in South Africa as evidenced by the largest desalination plant in the country at Mossel Bay [20]. According to regulations, a permit must be acquired to discharge large amounts of waste into the ocean [21], but the precedent from Mossel Bay demonstrates that South Africa will grant such a permit to a water desalination plant. In addition, research of nearby protected marine environments was conducted, and it was concluded that the proposed plant location near Amanzimtoti is at least 25 km from the nearest Marine Protected Area [22]. Therefore, direct brine disposal is the chosen alternative for the process.
Process Overview
Raw seawater is taken from the ocean and pumped to the pretreatment facility. Sodium hypochlorite is added to remove particulates and biological organisms that would cause fouling in the RO membranes. Pretreatment involves a series of filters to remove the majority of the particulates [13] and an activated carbon filter to dechlorinate the pretreated water before it is pumped to high pressure. Reverse osmosis occurs in two banks of spiral wound membrane modules. The high pressure concentrate exits the reverse osmosis system and is sent to an energy recovery device. This converts mechanical energy from the pressurized concentrate into mechanical energy that pumps the low pressure filtered seawater. The low pressure concentrate is then returned to the ocean. The purified product from reverse osmosis is sent to posttreatment where it is remineralized by mixing with a slight percentage of pressurized concentrate in order to restore the necessary minerals and to meet drinking water standards. It is then disinfected with chlorine dioxide and sent to the consumer. See Figure 2 for a block flow diagram of this process.
Process Flow Diagram
See Appendix G for the full process flow diagram. The process has been divided into two sections to aid in organization. The pretreatment section is represented by the 100 series of equipment while the reverse osmosis and posttreatment section is represented by the 200 series. A detailed layout of the reverse osmosis modules is provided in Figure G3 in Appendix G.
In the pretreatment step, a cascade of filters is implemented in the following order: macrofiltration, microfiltration, activated carbon, ultrafiltration and nanofiltration. Three pumps are utilized to account for the pressure drops across these filters. After pretreatment, the clarified seawater stream is split and sent to a pump and an energy recovery device to pressurize the reverse osmosis feed. After going through both reverse osmosis banks, the deionized water is remineralized with brine and disinfected with chlorine dioxide. The brine is used in the energy recovery device, then returned to the sea.
Control systems were added to this process to maintain safe operations and a standard level of product quality. For pretreatment, a fail-close system measures the level of seawater in V-100 and regulates the amount of sodium hypochlorite needed to disinfect the seawater. In the 200 series, two control loops were implemented, including a fail-close pressure control loop that maintains the necessary pressure by monitoring the concentrate entering C-200. A fail-open level controller measures the amount of purified water stored before post-treatment. The valve can increase the inlet flow rate of the final storage tank if the liquid level of purified water exceeds the set-point. This by no means encompasses all the control that should be implemented in the process and only serves as a representation of the necessity of these systems.
Mass and Energy Balances
The feed stream is assumed to be at the average seawater temperature of around 25℃ and at 1 atm [23]. The inlet water stream is estimated to have 500 ug/L of particulate matter. Flow rates, conditions, and compositions of each stream can be found in the stream table in Appendix H.
Pressure drops calculated for each pump and filtration step to estimate the utility requirements in the process can be found in Appendix I. The Ergun equation was used to calculate the pressure drops across each level of macrofiltration, which removes all particulate matter. Imperfect packing was assumed, with a void fraction of 0.4. The particle diameter was based off of the Wentworth Grain Size Chart [24], which can be found in Appendix J. The diameter of the filtration tank is 3.5 m because having a larger cross-sectional area decreases velocity. The pressure drops for the microfiltration, ultrafiltration, and nanofiltration modules were calculated using the Darcy equation and transmembrane pressure relationships from literature [25].
Granular activated carbon (GAC) filtration will be used for the dechlorination of the pretreated water. According to the US EPA, surface loading rates for GAC filters range between 2 to 10 gpm per square foot [26]. Using this typical flow rate, the pressure drop of flow through the activated carbon filter was estimated to be 0.6 psi per foot of bed depth found in Appendix K [27]. The activated carbon filter is assumed to effectively remove all of the hypochlorite from the stream.
The amount of brine used to remineralize the permeate ensures that the chlorine levels of the drinking water outlet remain below the upper limit set by South African regulations, as stated in Appendix C. The required concentrations of active chlorine in pretreatment and posttreatment to effectively disinfect the streams are 3 mg/L and 0.2 mg/L, respectively [28, 29].
Design Constraints
Sustainability has been a strong motivator throughout the design process. An energy recycle system has been implemented to combat this issue. This allows for cheaper production cost and lower energy requirements. Further sustainability efforts could include utilizing renewable energy sources such as solar, wind, and nuclear power. Environmental and ethical considerations are involved with releasing brine back into the sea. Although there are no laws controlling the release of concentrate back into the ocean, the process has been designed to carefully consider the impact and location of the release. The process avoids dangerous effects of this action by being located in an area far from declared marine protected areas. In addition, the brine will be released 500 m away from shore, decreasing its impact on coastal marine life.
The most pressing ethical consideration is the required level of water quality. By selecting a location outside of the United States, different regulations and standards could be applied to the purified drinking water that will be sent to consumers. The drinking water produced in this process will not only meet the drinking water standards of South Africa but will also exceed those limits to provide a healthier and cleaner water alternative allowing for a better drinking water experience.
Throughout the plant, safety considerations heavily impact the specifics of the process. Multiple control loops were implemented to ensure effective and safe production. The chloride concentration is near its maximum possible level. If the chloride ion concentration increases, it could cause increased corrosion. Therefore, to ensure a longer lifetime for the process, all pipes in the system are to be made with polyethylene. An increase in the corrosiveness of the water could cause metals ions to contaminate the water with certain types of piping as is currently being seen in Flint, Michigan. Therefore, chloride ion concentration will be carefully monitored to prevent negative health effects.
Equipment Sizing and Trade-offs
Equipment was sized to ensure that capital costs are not unreasonably high while satisfying the design basis requirements. The macrofiltration tank is filled with large amounts of sand with different levels of coarsity. The tank was designed to be as wide as possible at 3.5 meters to minimize the pressure drop and pumping requirements. For the amount of water being filtered daily, roughly 50 kg of particulate matter will accumulate. At an approximate density of 3,000 kg/m3, this would result in an accumulation of 16.7 liters of particulate matter per day. Thus, with a total height of 2 meters, there should be enough space to accommodate both sand and accumulated particulate matter.
Filters are sold as compact modules containing a large surface area. Increasing the area of the filters decreases the pumping requirements of the system, but increases the capital cost of purchasing the required filters. For each filtration step, many of these units will be used in parallel to meet the filtration surface area requirements, shown in Appendix L. Care was also taken to ensure that the pressure drop across the filters did not exceed product specifications.
The system incorporates three storage tanks. The first tank has a inlet flow rate of 7080 m3/hr, and the second and third tanks each have inlet flows of 5670 m3/hr. Because these flow rates are high, the tanks cannot hold water for long periods of time. The storage capacity for this facility was chosen to be 1.5 hours’ worth of flow. With two parallel units at each step to ensure repairs can be made efficiently, the total storage capacity of this facility will be about 64,000 m3 [30].
Dead end filters were chosen for all of the filtration steps. This increases the capital cost required as the cleaning process for dead end filtration necessitates the purchase of an excess of membranes. Filters will be active for 60 seconds and subsequently cleaned for 5 seconds while the flow is rerouted to another set of membranes. During cleaning, air is sent through the filters in the direction opposite the flow. Purchasing an excess of filters ensures that the required surface area is always available even though roughly 1/13th of all filters will be undergoing cleaning at any moment [31]. The benefit of dead end filtration is that it results in a higher yield, and therefore lower pumping requirements overall.
Economic Analysis
The current capital cost for the project is $22.4 MM, with an OSBL capital cost of $8.96 MM, as predicted by the Aspen Process Economic Analyzer. This capital cost includes the equipment cost, $18.4 MM, the membrane costs for pretreatment and reverse osmosis, $2.1 MM, and piping, land, and facility costs totaling $1.9 MM. Detailed breakdown of equipment and membrane costing can be seen in Appendix M. Equipment costs have been estimated using fiberglass reinforced plastic for construction of tanks and using polyethylene for piping to reduce corrosion [32]. Periodic backwashing is required to remove any particulates that gather during filtration; this cost has been accounted for in maintenance, which was estimated to be 7% of the ISBL investment [33, 34].
The main revenue of producing approximately 132,000 m3 per day of desalinated water sold at $0.81 per metric ton is $37.5 MM [35]. Utility costs for this design are $13.7 MM per year. This value arises from the requirement of 20,209 KW of electricity purchased at $0.08 per KWh [36]. To decrease the environmental impact from the large utility requirement, electricity will be obtained from the Ingula Pumped Storage Scheme [37], a hydropower plant located approximately 250 km away from our proposed location.
Raw material costs for this project are relatively low, approximately $0.04 MM. This is mainly due to the fact that the seawater is obtained less than 500 m away from the coast and does not require transport or processing. The sodium hypochlorite for chlorination during pre-treatment can be purchased at $108 per metric ton [38], and the chlorine dioxide can be purchased at approximately $2200 per metric ton [39].
Land cost was estimated using online property listings by finding different vacant lots near Amanzimtoti. The cost per acre was estimated to be R550,000 or $35,000. The required land area is approximately 8 acres [40], resulting in total land costs of approximately $300,000. The predicted salary and overheads are $0.8 MM, which include 3 shifts of 5 operators at a salary of R150,000 or $9,500 per operator, the average salary for a plant operator in South Africa [41].
Using typical industrial economic relationships, the OSBL was estimated to be 40% of the ISBL, the engineering costs to be 30% ISBL plus OSBL, the contingency charge to be 10% of the ISBL plus OSBL, and working capital to be 20% of the total fixed capital cost. The construction schedule models plant building during the first two years at 40% and 60% investment, respectively. Full production is set to begin immediately upon plant completion. The depreciation rate chosen was the 5 year MACRS method because it yields better return than the straight-line model. The tax rate was selected to be 35%, reflecting the standard industrial tax rate in South Africa [42].
The net present value at 10 years is predicted to be -$34.2 MM and increases to -$23.5 MM at 20 years with an internal return rate is 2.9% at 20 years. As of now, the process is not economically feasible. However, as described by the sensitivity analysis below, the process may become economically viable if utility costs and product revenue change favorably.
Optimization and Sensitivity Analysis
A cost optimization was performed to determine how reverse osmosis modules could be organized to minimize costs. As the number of modules increases, the capital and operating costs decrease as seen in Figure 3. The module costs are trivial in comparison to the capital costs of the pumps. As the number of modules approaches 900, utility requirements approach a minimum value, but the number of modules needed in the second bank begins to increase exponentially, causing a spike in the capital costs. As a result, the optimal balance between operating costs and utility costs was found to be 900 modules in the first bank and 4000 modules in the second bank.
A sensitivity analysis was performed to elucidate the effect of four major variables on the NPV at the end of 20 years: selling price of water, maintenance cost, operating cost, and capital cost. Each variable was modified between 75% and 125% of its present value to determine its effect on the NPV. Overall, the price of water has the largest impact, followed by the operating costs, capital costs, and the maintenance costs, as seen in Figure 4.
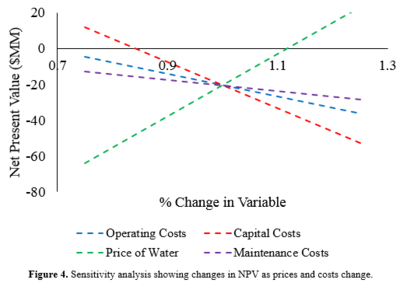
Although the NPV is currently negative, the sensitivity analysis shows a path towards having a positive NPV. If the price of water increases by 15% and the cost of energy decreases by 15%, the NPV reaches a positive value. These are values that could potentially be reached through negotiations with various government agencies and energy providers. It may be possible to obtain electricity at a lower price due to the construction of a 3,000 MW hydroelectric dam in 2017. Additionally, due to the rising water scarcity in Africa, an increase in the price of water can be expected in the future.
Conclusion
The proposed process meets the design requirements of producing 100,000 m3 per day of clean drinking water. However, due to high operating costs and significant capital costs, the design is not economically viable, due to a 20 year NPV of -$23.5 MM. A sensitivity analysis showed that the price of water is most important for determining profitability. Currently, the selling price of water is too low for the plant to be profitable. If the plant were to be constructed, contracts could be negotiated between suppliers and electricity providers in order to provide a consistent lower price. Therefore, it is not recommended to implement the plant design unless there are significant changes in the market for drinking water in South Africa.
References
- International Decade for Action 'Water for Life'. UN News Center. Available at: http://www.un.org/waterforlifedecade/scarcity.shtml. Accessed January 13, 2016. This source was used to find statistics about water scarcity to better understand the problem.
- Greenlee LF, Lawler DF, Freeman BD, Marrot B, Moulin P. Reverse osmosis desalination: water sources, technology, and today's challenges. Water Res. 2009;43(9):2317-48. This source contains specifics about the reverse osmosis process and similar technologies.
- United Nations Environment Programme. Increased global water stress. 2008. http://www.unep.org/dewa/vitalwater/article141.html
- Madison Powers. Water Scarcity. 2015. http://www.fewresources.org/water-scarcity-issues-were-running-out-of-water.html
- Humphreys, K.K. Sources of International Cost Data. Presented at NORDNET ‘97 Conference. Reykjavik, Iceland. September 11, 1997.
- Water purification plant solutions. Veolia. Available at: http://www.veoliawaterst.co.za/municipal-water-treatment/water-purification/water-purification-plant/. Accessed January 13, 2016. This source was used to determine locations for other desalination plants in South Africa.
- Map of ocean salinity. Science Learning Hub RSS. Available at: http://sciencelearn.org.nz/contexts/the-ocean-in-action/sci-media/images/map-of-ocean-salinity. Accessed January 13, 2016. This source was used to classify the salinity of the water surrounding South Africa.
- IDE's Israel seawater RO desalination plant sets world record for water production. - WaterWorld. Available at: http://www.waterworld.com/articles/2015/02/ide-s-israel-seawater-ro-desalination-plant-sets-new-world-record.html. Accessed January 15, 2016. This source was used to determine the water intake of the greater city of Durban for plant capacity sizing.
- Li WKW. Annual average abundance of heterotrophic bacteria and Synechococcus in surface ocean waters. Limnology and Oceanography 1998;43(7):1746–1753. Available at: http://www.aslo.org/lo/toc/vol_43/issue_7/1746.pdf. Accessed January 15, 2016. This source was used to determine the concentration of bacteria in seawater.
- Pearson - Science News. Pearson - Science News. Available at: http://www.phschool.com/science/science_news/articles/all_worlds_phage.html. Accessed January 15, 2016. This source was used to determine the concentration of viruses in seawater.
- Holmes S. South African Water Quality Guidelines. 2nd ed. Pretoria: The Dept. of Water and Forestry; 1996.
- Water Treatment Solutions. (n.d.). http://www.lenntech.com/processes/desalination/pretreatment/general/desalination-pretreatment.htm Accessed January 22, 2016 This source was used to determine pretreatment for RO.
- Sagle A, Freeman B. Fundamentals of Membranes for Water Treatment. The Future of Desalination in Texas. 2004;2. http://www.twdb.texas.gov/publications/reports/numbered_reports/doc/r363/c6.pdf Accessed January 28, 2016. This source provides information about different types of membranes for RO filtration.
- Membrane Technologies. Koch Membrane Systems. http://www.kochmembrane.com/Learning-Center/Technologies.aspx Accessed January 28, 2016. This source provides the effectiveness of RO membranes.
- Desalination Post-treatment: Neutralization/Remineralization for Drinking Water. Lenntech. http://www.lenntech.com/processes/desalination/post-treatment/post-treatments/remineralization.htm Accessed January 20, 2016. This source provides a description of different remineralization systems.
- Disinfection. Lenntech. http://www.lenntech.com/processes/disinfection/disinfection.htm Accessed January 29, 2016. This source provides options for disinfection.
- Ocean Standards. California Environmental Protection Agency: State Water Resources Control Board. http://www.waterboards.ca.gov/water_issues/programs/ocean/desalination/ Accessed January 20, 2016. This source provides the impact of brine on the environment.
- Brandhuber P, Cerone J, Kwan P, Moore E, Vieira A. A look at conventional and emerging brine disposal and waste minimization technologies. HDR Waterscapes. 2008; 9; 2. https://www.hdrinc.com/sites/all/files/content/articles/article-files/3601-a-look-at-conventional-and-emerging-brine-disposal-and-waste-minimization-technologies.pdf Accessed January 20, 2016. This source provides an overview of the alternatives available for brine disposal.
- Tularam GA, Ilahee M. Environmental concerns of desalinating seawater using reverse osmosis. Journal of Environmental Monitoring. 2007; 9; 805-813. http://pubs.rsc.org/en/content/articlehtml/2007/EM/B708455M Accessed January 27, 2016. This source states that discharging back into the ocean is the most common method of disposal.
- South Africa’s Largest Seawater Desalination Plant. Veolia Water Solutions & Technologies. http://www.veoliawaterst.co.za/vwst-southafrica/ressources/files/1/32048,Mossel-Bay-Desalination.pdf Accessed January 27, 2016. This source provides information on the desalination plant at Mossel Bay.
- Department of Environmental Affairs. National Guideline for the Discharge of Effluent from Land-based Sources into the Coastal Environment. 2014. https://www.environment.gov.za/sites/default/files/legislations/nationalguideline_landbasedinfluent_dischargecoastal.pdf Accessed January 27, 2016. This source provides information on obtaining a permit for discharging effluent back into the ocean.
- Tunley K, Chadwick P, Nobula S. State of Management of South Africa’s Marine Protected Areas. WWF South Africa Report Series. 2009. http://assets.wwfza.panda.org/downloads/mpastateofmanagementreport04nov2009weblowerdpi.pdf Accessed January 27, 2016. This source provides locations of marine protected areas near South Africa.
- Global Sea Temperatures - Durban Average Monthly Sea Temperature. Sea Temperature. http://www.seatemperature.org/africa/south-africa/durban-february.htm Published 2016. Accessed February 18, 2016.
- U.S. Geological Survey. Surficial Geology of the Sea Floor in Long Island Sound Offshore of Orient Point, New York. USGS. http://pubs.usgs.gov/of/2010/1100/html/fig10.html Accessed February 18, 2016.
- Crittenden J. Water Treatment: Principles and Design. Wiley; 2005.
- Drinking Water Treatability Database. Granular Activated Carbon. U.S. Environmental Protection Agency. http://iaspub.epa.gov/tdb/pages/treatment/treatmentOverview.do?treatmentProcessId=2074826383 Published 2016. Accessed February 18, 2016.
- Clack Corporation. Granular Activated Carbon (GAC). http://www.farriswater.com/pdf/gac.pdf Published August 13, 2008. Accessed February 18, 2016.
- Lenntech. Chemical Disinfectants - Chlorine. http://www.lenntech.com/processes/disinfection/chemical/disinfectants-chlorine.htm Accessed February 19, 2016.
- Lenntech. Chemical Disinfectants - Chlorine Dioxide. http://www.lenntech.com/processes/disinfection/chemical/disinfectants-chlorine.htm Accessed February 19, 2016.
- Alibaba.com. Building fiberglass cooling water storage tank. http://www.alibaba.com/product-detail/building-fiberglass-cooling-water-storage-tank_60019235269.html?spm=a2700.7724838.30.119.kzLzuC Accessed Feb 27, 2016.
- Lenntech. Membrane Cleaning Methods. http://www.lenntech.com/membrane-cleaning.htm Accessed Feb 29, 2016.
- Alibaba.com. Underground water large diameter plastic drip irrigation pipe. http://www.alibaba.com/product-detail/underground-water-large-diameter-plastic-drip_60213689907.html?spm=a2700.7724838.0.0.NrgLIM Accessed Feb 29, 2016.
- DeSilva, F. Water Treatment Guide. Activated Carbon Filtration. http://www.watertreatmentguide.com/activated_carbon_filtration.htm Published January 2000. Accessed Feb 29, 2016.
- Muilenberg, T. Water and Wastes Digest. Microfiltration: How does it compare? http://www.wwdmag.com/desalination/microfiltration-how-does-it-compare Published December 28, 2000. Accessed Feb 29, 2016.
- Sweet, P. Las Vegas Sun. Desalination Gets a Serious Look. http://lasvegassun.com/news/2008/mar/21/desalination-gets-serious-look/ Published March 28, 2008. Accessed Feb 29, 2016.
- Business Tech. Electricity Prices: South Africa vs the World. http://businesstech.co.za/news/energy/99494/electricity-prices-south-africa-vs-the-world/ Published September 2015. Accessed Feb 28, 2016.
- Eskom. Ingula Pumped Storage Scheme. http://www.eskom.co.za/Whatweredoing/NewBuild/IngulaPumpedStorage/Pages/Ingula_Pumped_Storage_Scheme.aspx Accessed Feb 27, 2016.
- USP Technologies. Sodium Hypochlorite. http://www.h2o2.com/products-and-services/us-peroxide-technologies.aspx?pid=116 Published 2016. Accessed Feb 29, 2016.
- www.proformindustries.com/~proform/sites/.../purewater_presentation.pptx
- Tampa Bay Water. Seawater Desalination Plant Data Sheet. http://www.tampabaywater.org/documents/fact-sheets/desal-fact-sheet.pdf Published 2010. Accessed Feb 27, 2016.
- PayScale Human Capital. Average Salary for People with Jobs as Plant Operators/Managers/Supervisors. http://www.payscale.com/research/ZA/People_with_Jobs_as_Plant_Operators%2FManagers%2FSupervisors/Salary Accessed Feb 29, 2016.
- Trading Economics. South Africa Corporate Tax Rate. http://www.tradingeconomics.com/south-africa/corporate-tax-rate Accessed Feb 27, 2016.
Appendices
Appendix A
This appendix includes a map representing the salinity of seawater around the world in parts per thousand.
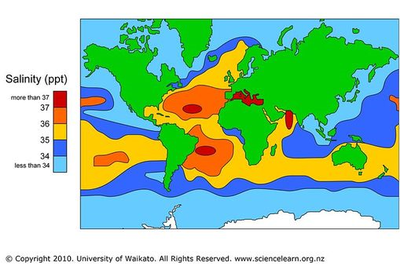
Appendix B
This appendix includes the locations of the five existing desalination plants in South Africa.
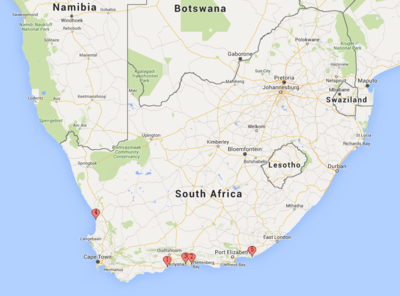
- Knysa - Western Cape
- Plettenberg Bay
- Mossel Bay
- Lamberts Bay
- Cannon Rocks and Boknes Communities
Appendix C
This appendix includes the inorganic compositions in seawater along the coast of South Africa, as well as the upper limits for each of these species.
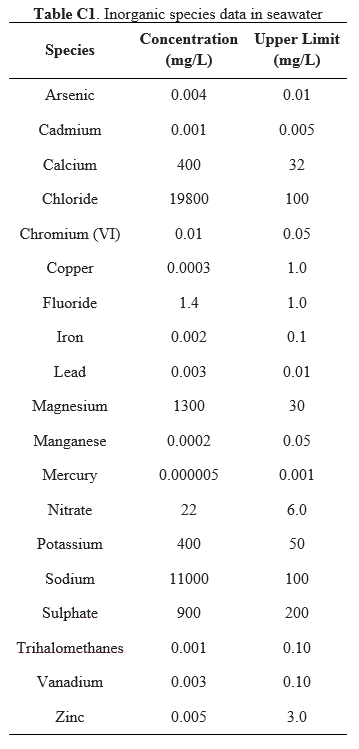
Appendix D
This appendix provides summaries of process alternatives for pretreatment.

Appendix E
This appendix includes the different types of reverse osmosis membrane modules.
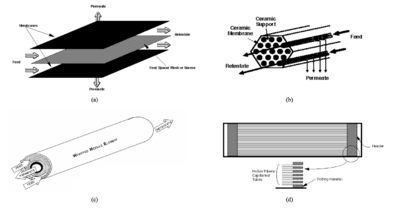
Appendix F
This appendix includes the different alternatives for remineralization and the included comparison of capital and operating costs.
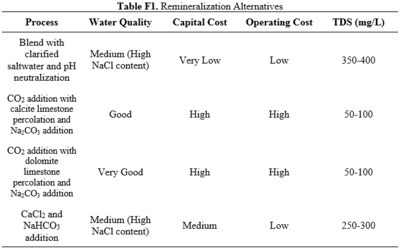
Appendix G
This appendix contains the process flow diagrams divided into the 100 section for pretreatment and the 200 section for reverse osmosis and posttreatment. A layout of the banks for reverse osmosis is also included.
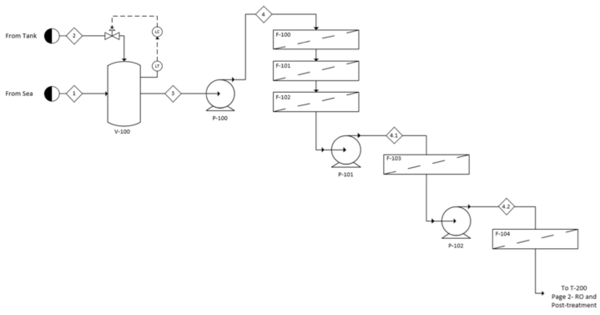
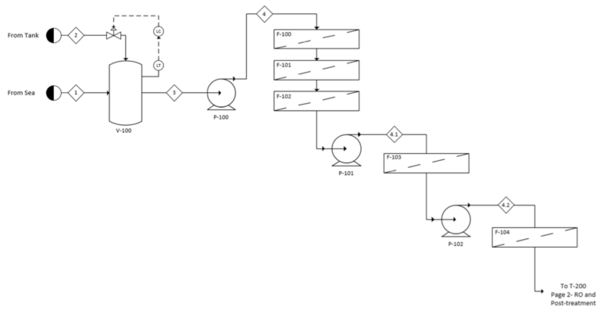
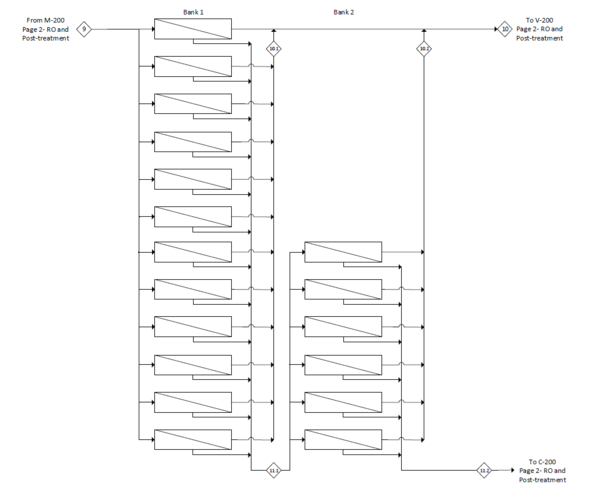
Appendix H
This appendix includes stream conditions and composition in the process.
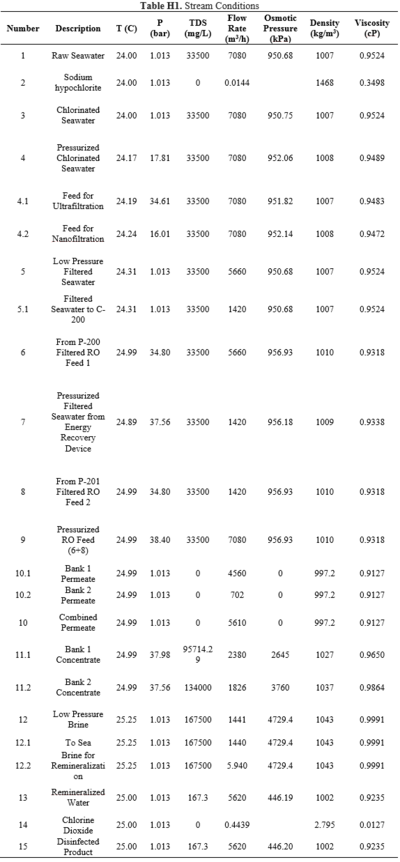
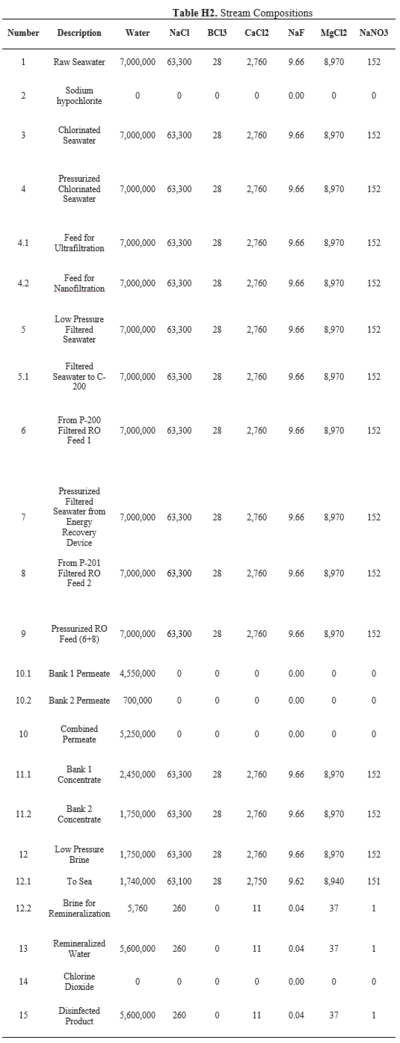
Appendix I
This appendix includes the energy requirements for each pump in the process.

Appendix J
This appendix includes the Wentworth Grain Size Chart used for pressure drop calculations of the macrofiltration step in pretreatment.

Appendix K
This appendix includes the calculated pressure drops across all the membranes used in the process.
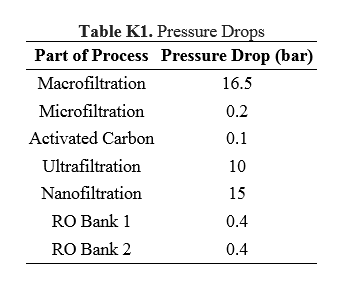
Appendix L
This appendix includes the results of the surface area calculations for each step in the process.
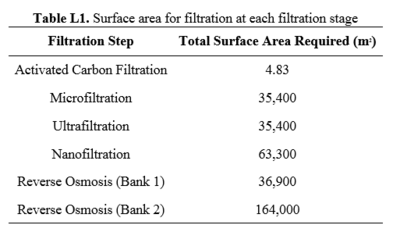
Appendix M
This appendix includes the economic analysis spreadsheet, along with the costs of production and a graph showing the resulting net present value with changing the number of reverse osmosis membranes in the first bank of the system.