Reactor: Difference between revisions
/* HYSYS ReactorsG.P. Towler, R. Sinnott, Chemical Engineering Design: Principles, Practice and Economics of Plant and Process Design. p.186-194, Elsevier (2012).AspenTech. HYSYS 2005.2 Simulation Basis. Chapter 9 (2005).Rice University Chemical Engine... |
|||
| Line 110: | Line 110: | ||
==Simulation== | ==Simulation== | ||
===Degrees of Freedom | ===Degrees of Freedom=== | ||
A degree of freedom analysis will assist in reaching a state of convergence for the reactor and downstream units. For a reactor, | A degree of freedom analysis will assist in reaching a state of convergence for the reactor and downstream units (Felder, 2005). For a reactor, | ||
<math>N_{dof}=N_{unknowns}+N_{reactions}-N_{balances}</math> | <math>N_{dof}=N_{unknowns}+N_{reactions}-N_{balances}</math> | ||
in which the number of degrees of freedom is expressed as the number of unknown variables plus the number of reactions occurring minus the number of material balances able to be performed on the system. In order for the simulation to converge, the user must specify as many variables as existing degrees of freedom. The simulation will then calculate unspecified variables. | in which the number of degrees of freedom is expressed as the number of unknown variables plus the number of reactions occurring minus the number of material balances able to be performed on the system (Wikibooks, 2014). In order for the simulation to converge, the user must specify as many variables as existing degrees of freedom. The simulation will then calculate unspecified variables. | ||
==Conclusion== | ==Conclusion== | ||
HYSYS simulations are an excellent starting point for designing a plant; but, they can also be used after completion to check for accuracy in the sizing and rating of the equipment. This will help to ensure that a plant is safe for employees and will have safety precautions in place to prevent dangerous incidents. | HYSYS simulations are an excellent starting point for designing a plant; but, they can also be used after completion to check for accuracy in the sizing and rating of the equipment. This will help to ensure that a plant is safe for employees and will have safety precautions in place to prevent dangerous incidents. | ||
Revision as of 01:25, 9 March 2015
Authors: Vincent Kenny [2015] and Stephen Lenzini [2015]
Steward: Jian Gong and Fengqi You
Introduction
Process simulation is extremely beneficial to engineers, allowing them to further understand processes, identify process advantages and limitations, and provide quantitative process outputs and properties. Modeling reactors and their corresponding reactions is difficult by nature but can be rewarding if done correctly. This page provides essential information on the topic of reactor simulation using the computer program Aspen HYSYS.
Aspen HYSYS Reactor Simulation Basics
The HYSYS program allows the user to define reactions primarily based on desired model outputs and available information. After defining process components, the user can choose a reaction type as listed in the section below. HYSYS includes a number of different reactor models for the various reaction types, desired outputs, and specification limitations as well as standard PFR and CSTR models as described in this section.
Unlike other simulation programs, HYSYS attempts to calculate all variables at all times which can be helpful or frustrating at times. The user can utilize the "hold" function in order to cease this process.
Limitations
Because simulation requires reaction characteristics, parameters, and other information, it is important to conduct background research appropriate to the reaction of interest before beginning the actual simulation. If theoretical or empirical data do not exist for the reaction, it may be difficult or impossible to conduct a computer simulation (see Additional Options). It follows that an increasing number of reaction byproducts requires an increasing amount of reaction data. Users should not be concerned if they cannot quantitatively specify or simulate all reaction by-products or outputs; in the end, a simulation as a process approximation and is inherently unable to model the process completely.
Of course, the phase of the reaction must be known; unfortunately, however, HYSYS does not support solid phase modeling and thus a different approach must be chosen (AspenTech).
Research
When dealing with chemical reactions, knowing details about the reaction is key. The products and reactants should be known and any identifying properties known. Examples of the properties being referred to are the phase of the reaction, the interactions between molecules, and the effects of the reaction occurring. In addition, any potential side reactions that can occur should be noted and the conditions under which they are more likely to occur should be identified.
Phase
HYSYS is able to simulate solids, liquids, and gases. When working exclusively with gases and liquids, there is little information required other than operating parameters. Using the fluid package specified and equilibrium data, HYSYS will determine the vapor and liquid fractions. When modelling solids, additional information is required. Because solids are more heterogeneous than liquids and gases, information such as the size of the particles used, chemical composition, and moisture content must be input into a simulation (AspenTech).
Fluid Package
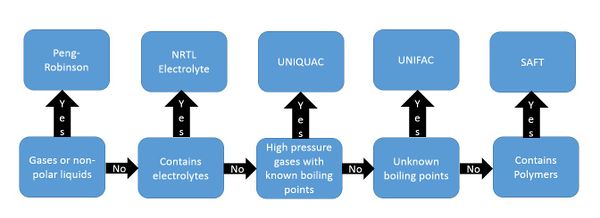
There are numerous fluid packages to choose from in HYSYS, however most simulations can be accomplished with one of five fluid packages. If the system is entirely gases or nonpolar liquids, then Peng-Robinson will work well. If it contains electrolytes in the reaction, NRTL electrolyte will be effective at simulating the reaction. If simulating high pressure gases where the boiling points of the components are known, UNIQUAC will accurately simulate the system; if the boiling points are unknown UNIFAC may work better. If the reaction contains polymers, the SAFT fluid package is able to account for the properties in the reaction (Washington State University).
HYSYS Reactions
Before entering the simulation environment, certain steps must be taken to ensure that HYSYS is able to create a model that will be useful. A component list must be created, the reactions occurring, including unwanted reactions, should be noted, and the fluid package must be specified, and the type reaction to be simulated must be selected.
Components
The input components must be selected that will be reacting. To do so, go to the tab labeled as component list. There is the option to add a new component. The components can be searched for by either their scientific or common names. It is not necessary to select the products of the reaction at this step and HYSYS will be able to determine the products. HYSYS is also able to determine any intermediate components that must be created during a reaction.
Conversion Reaction
A conversion reaction is the simplest type in HYSYS. A conversion reaction operates on a stoichiometric basis and will run until either the limiting reagent is exhausted or until the specified conversion has been achieved. These reactions do not function well with other reactions occurring simultaneously and should be used exclusively with other conversion reactions (Reactions in HYSYS). When specifying the conversion fraction it is important to use the percent form rather than the decimal form; meaning input 50 rather than .5 for half conversion.
Equilibrium Reaction
Equilibrium reactions require more inputs than conversion reactions do. They rely upon the reactions equilibrium constant, Keq. There are many different ways to input Keq into HYSYS. The first method is to assume that the equilibrium constant has a set value and input the desired value. The next method is to use a relationship to determine the equilibrium constant as a function of temperature and tabulate a series of equilibrium constants over the applicable temperature range. HYSYS is able to generate equilibrium constants for systems that behave in an ideal manner using the Ideal Gas Gibbs Free Energy Coefficients. Equilibrium reactions should be performed in the Equilibrium Reactor, General Reactor, or Gibbs Reactor although the stoichiometry will be a factor when using the Gibbs Reactor. The simulation is not accurate or useful when performed in a CSTR or PFR (Reactions in HYSYS).
Heterogeneous Catalytic Reaction
As the name suggests, the Heterogeneous Catalytic Reaction pack is used to simulate systems with a catalyst. The catalyst used in these reactions is most frequently a solid and the porosity, void fraction, and the chemical makeup and moisture content should be specified. The simulation is most effective when used in a PFR reactor as it is the most likely scenario where a solid catalyst is used although it can also be simulated in a CSTR but this has the potential to damage the equipment when the impellers strike the catalysts (Reactions in HYSYS).
Kinetic Reaction
Kinetic reactions require an input of the reaction parameters. A kinetic reaction depends on the parameters unique to the system and the concentration of the reactants as well as the products. The parameters required are the reaction constants, k, activation energies, E, and the pre-exponential factors, A. The reaction order is input by the user so that HYSYS may know how to treat the reaction as a function of the concentrations. This reaction type also ahs the possibility for a backwards reaction, however, this will only occur if the activation energy and pre-exponential factors have been input for the reverse reaction. Kinetic reactions are best simulated using a CSTR or a PFR (Reactions in HYSYS).
Managing Reactions
When managing reactions, the reactions that will occur must be specified in stoichiometric balance. This step is key to the simulation being successful because if a reaction should occur and is not listed, HYSYS will not have that reaction occur. In addition, this step is how the product of a reaction are inserted into the simulation environment as they were not added in the component list. This is also the step where any reaction parameters are input into HYSYS. A shortcoming of early HYSYS versions is that only two significant figures will be displayed even when additional digits are included making it potentially confusing if sharing a simulation with others. The other convention of not for HYSYS reaction management page is that the Reaction Heat is defined such that exothermic reactions are positive, contrary to how heat of reaction is usually defined (Reactions in HYSYS).
HYSYS Reactors
In general, the user will attach a defined reaction set, generated as described above, to each reactor. This allows precise coordination of reactions that may be occurring in different units in the entire process.
Plug Flow Reactor (PFR)
Plug Flow Reactors are tubular reactors that assume perfect radial mixing and approximate zero axial dispersion (Towler, 2012). A PFR is used primarily for gas phase reactions. HYSYS PFRs can utilize kinetic, equilibrium, and heterogenous catalysis reaction types. The user must define an inlet streams and an outlet stream; like most HYSYS units, multiple streams can enter the same unit. An optional energy stream can be specified to either provide or remove heat; if no stream is specified, HYSYS assumes that the unit operates adiabatically. However, depending on user specifications, HYSYS can determine required energy for the specified operation.
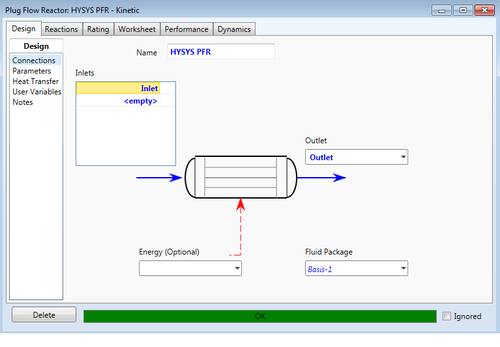
The user can view parameters and variables resulting from the integration of physical specifications and the specified chemical reaction after the operation converges.
Unique Parameters
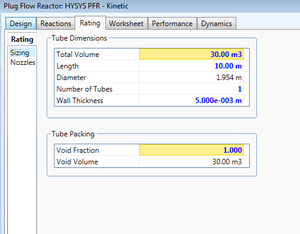
The user must define physical properties of the reactor such as volume, cross sectional area, and length. Based on geometry, only two of the three parameters must be specified. Additionally, the program provides the option to define a number of tubes included in the unit. The specified length will encompass all tubes.
Pressure drop through the PFR can be defined by the user, approximated as zero, or left for HYSYS to calculate. User-defined pressure drops can be useful in accordance with the process as a whole but a zero approximation can serve for initial simulations if a pressure drop is not know. HYSYS will calculate using the Ergun equation.
A void fraction must be specified with unity referring to zero reactor packing. If a value other than unity is specified, the user must input data for the packing. Note that this data has a purely physical relationship with the system such as mean particle size, solid density, solid heat capacity, etc. and does not relate the the chemical reaction occurring in the unit. If the PFR is using a reaction set with a Heterogeneous Catalytic Reaction, that information will be specified in the reaction itself.
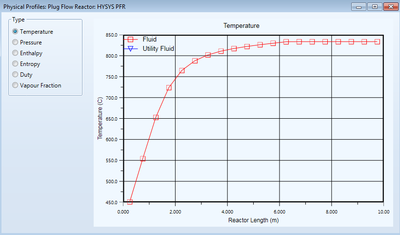
Sub-volume Integration
The HYSYS PFR divides the unit into sub-volumes based on total length where calculations occur throughout the PFR. The default number of sub-volumes is 20, but this number can be specified by the user. Of course, a higher number of sub-volumes will generally lead to a more accurate simulation but may be unnecessary given the operation or limitations due to time and computation capacity. Properties of unresolved sub-volumes can be user-specified using the "Minimum Step Fraction" and "Minimum Step Length" inputs. The Performance tab can provide useful information about various variables and their values in each sub-volume. Additionally, after convergence, the user can view various graphs of process variable changes throughout each sub-volume.
Continuous Stirred Tank Reactor (CSTR)
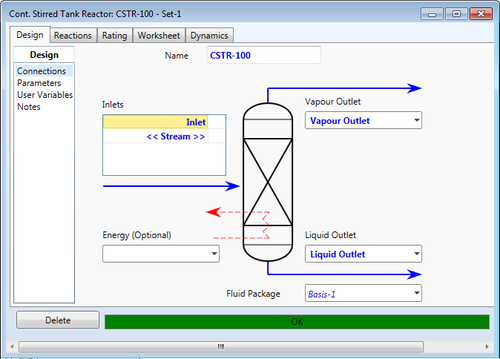
The Continuous Stirred Tank Reactor assumes perfect mixing throughout the volume of the reactor and the outlet conditions are approximated as equal to conditions inside the reactor. The "General Reactors" (Equilibrium Reactor, Conversion Reactor, Gibbs Reactor) each is a CSTR with a specialization in its corresponding HYSYS Reaction Type. The kinetics, equilibrium, and heterogeneous catalysis reaction types can be used with the generic CSTR.
The CSTR requires at least one input stream and can consider both vapor and liquid phases internally with corresponding liquid and vapor outlets. Primarily, CSTRs are used for liquid reactions, and thus the vapor outlet can be omitted, but the option exists nonetheless. The user can also omit the liquid outlet but this is not recommended and a PFR should be considered instead.
As with the PFR, an optional energy input can be specified or omitted to assume an adiabatic operation. Additionally, HYSYS can calculate the energy input given that the user specifies the appropriate variables.
A pressure drop is unlikely to be substantial with a CSTR but can be entered by the user; the default pressure drop is assumed to be zero, or HYSYS can calculate a pressure drop based on outlet stream information. The volume should be calculated or estimated and user-defined on the unit information page. Based on gemoetry, the user will need to enter two of the following: volume, diameter, or height. The default geometry is cylindrical, and the user can choose a verital or horizontal arrangement. An option exists to use a spherical CSTR as well.
Unique to the CSTR, an option exists to model as a flash drum separator if a solution cannot be found. This option may enhance or limit the desired outputs depending on the situation.
Like the PFR, after convergence, the CSTR unit page can show parameter and variable outputs based on integration of physical specifications and the chemical reaction.
Equilibrium Reactor
The Equilibrium Reactor is a CSTR with a specialization in equilibrium reactions and thus can only function with reaction sets including that reaction type. The outlets from this reactor will be in a state of chemical and physical equilibrium according to parameters defined in the reaction set; as a consequence, the operation is extremely sensitive to input parameters and variables. The user might find it easier to use the Gibbs Reactor in situations where sensitivity affects the program's ability to reach convergence.
Conversion Reactor
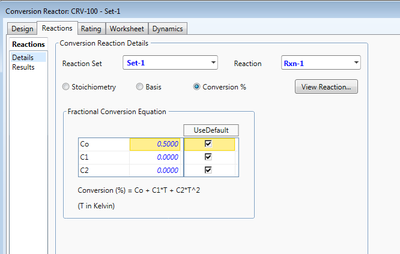
The Conversion Reactor is a CSTR with a specialization in conversion reactions and thus can only function with reaction sets including that reaction type. Unique to this reactor, conversion X becomes a process variable defined as
where A is the base component of the reaction. Like other variables, this can be solved for by HYSYS or specified by the user depending on the degrees of freedom analysis.
Conversion is defined through the base component specified when defining the reaction. The HYSYS conversion equation provides the capability to define conversion based on temperature to the first and second powers, for the second and third terms, respectively, of the equation. If those terms are set to zero, conversion will proceed as a constant value defined in the first term of the equation. In the figure, conversion is not temperature dependent and will convert 50% of the base component to products.
Due to the simplicity of this reaction type, this reactor can integrate multiple different reactions in the same unit. The product of one reaction can serve as the reactant of another, and so on. This can be extremely useful for complex reactor designs.
Gibbs Reactor
The Gibbs Reactor is an extremely unique CSTR in that it does not require a reaction set to be attached in order to function. The Gibbs Reactor will simply produce an outlet in which the Gibbs free energy of the mixture is minimized. Of course, a reaction set can be attached to this reactor, but it is important to note that any parameters specified in the set will not be included in the simulation as the minimization of free energy will be the dominant simulation method. An exception to this rule is the stoichiometry of the reaction set. Without a set, stoichiometry will not be considered in the simulation; however, with an attached reaction set with stoichiometric parameters, the simulation will account for them and the outlet conditions can change. However, if a set is attached, only the components specified in the set will reach an equilibrium point; other components will be neglected.
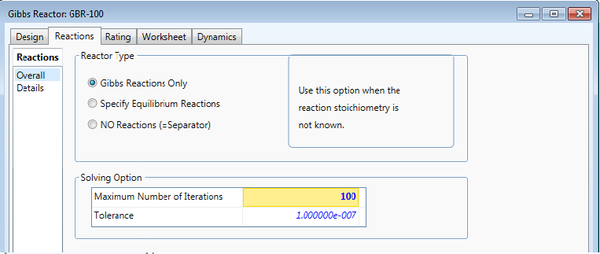
The "Reactions" tab on the Gibbs Reactor is where the user makes the decision described above. "Gibbs Reactions Only" minimizes the free energy of the inlet stream to produce the outlet streams. "Specify Equilibrium Reactions" allows the user to attach a reaction set in which free energy is minimized according to the equilibrium kinetics and stoichiometry of the set. Lastly, the reactor can serve as a flash separator by selecting "NO Reactions".
The Gibbs Reactor can be very useful if the user does not possess any data pertinent to the reaction or desires only a simulation of the equilibrium state. At the very least, the Gibbs Reactor can provide simulation estimates as a starting point for a more rigorous simulation through another reactor type.
Simulation
Degrees of Freedom
A degree of freedom analysis will assist in reaching a state of convergence for the reactor and downstream units (Felder, 2005). For a reactor,
in which the number of degrees of freedom is expressed as the number of unknown variables plus the number of reactions occurring minus the number of material balances able to be performed on the system (Wikibooks, 2014). In order for the simulation to converge, the user must specify as many variables as existing degrees of freedom. The simulation will then calculate unspecified variables.
Conclusion
HYSYS simulations are an excellent starting point for designing a plant; but, they can also be used after completion to check for accuracy in the sizing and rating of the equipment. This will help to ensure that a plant is safe for employees and will have safety precautions in place to prevent dangerous incidents.
Additional Options
In general, the AspenPlus simulation program can handle more rigorous process simulation. Several other types of simulation programs are available as well.
If reactor simulation through a program fails, an engineer can execute manual simulation for a unit by hand or through Microsoft Excel, Matlab, or another computational method. Because of the manual method, the results from this simulation should be more intuitively clear and can be added to the overall process on HYSYS absent an actual reactor and/or reaction.
References
AspenTech, "FAQ: Solids Modeling in AspenPlus", 2014
"Reactions in HYSYS" Rice University Chemical Engineering Department, [http://www.owlnet.rice.edu/~ceng403/hysys/reactions.html "Reactions in HYSYS"
Washington State University's Gene and Linda Voiland School of Chemical Engineering and Bioengineering, "Basics of HYSYS"
G.P. Towler, R. Sinnott, Chemical Engineering Design: Principles, Practice and Economics of Plant and Process Design. p.186-194, Elsevier (2012).
AspenTech. HYSYS 2005.2 Simulation Basis. Chapter 9 (2005).
Rice University Chemical Engineering Department. <http://www.owlnet.rice.edu/~ceng403/hysys/reactions.html>
R.M. Felder, R.W. Rousseau, Elementary Principles of Chemical Processes. 3rd edition, Wiley (2005).
Wikibooks, Introduction to Chemical Engineering Processes, 2014. <http://en.wikibooks.org/wiki/Introduction_to_Chemical_Engineering_Processes/Problem_considerations_with_molecular_balances>