Design S1
Design Report Authors: Michael Gleeson, Thomas Considine, Sean Kelton
Steward: Fengqi You
Date Presented: March 11, 2014
Executive Summary
The Chemical Intermediary Division ($1) of Evanston Chemical Corporation is interested in evaluating the economic benefit of adding 1,4 Butanediol (BDO) to its already robust offering of chemical intermediaries. It plans on producing 50,000 tonnes of 99.5%wt BDO using a modified Davy Process through a Gas Liquid Induction Reactor (GLIR) with a rare earth metal catalyst. The facility will be located in Lake Providence, LA , due to proximity to raw material productions sites and ease of transportation of the final product.
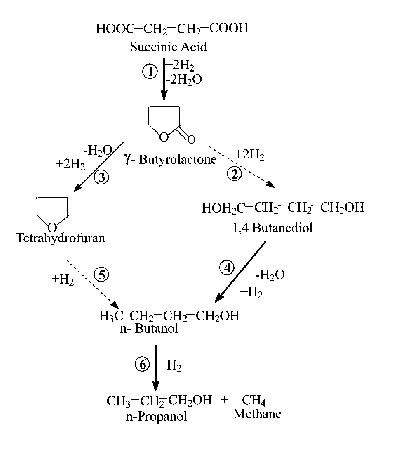
BDO is produced from Succinic Acid (SUC) by a hydrogenation reaction at very high pressures. This particular process uses a rare earth catalyst to because of the high conversion of SUC and the high selectivity toward BDO. Side products of the reaction include the highly valuable chemicals tetrahydrofuran (THF) and γ-butyrolactone (GBL), as well as water, n-butanol, and various other low value chemicals. The reaction occurs at 165oC and 150 bar, so special attention must be paid to reactor design to account for these extreme conditions.
Aspen HYSYS was used to model this process, and equipment sizes were calculated as specified by Towler. Equipment costs were estimated using Aspen Process Economic Analyzer (formerly known as Icarus). In order to meet the process specifications within the given time-frame, some simplifying assumptions were made that impose limits on the accuracy of the model; specifically, the GLIR was modelled using a conversion reaction. This means that the conversion remained unchanged at unideal operating conditions, which, clearly, is inaccurate. Additionally, only one reaction was modelled; in real world conditions, dozens of reactions would occur within the reactor, which would have a potentially massive effect on the accuracy of this model. Despite these limitations, the possibility of producing BDO for sale in the open market should be investigated because of the extremely encouraging economic potential of this endeavor.
Economic analysis of this process indicates a 10 year NPV of $163M, a 20 year NPV of $258M, and a total capital cost of $17.5M. The variable operational costs are approximately $131M. At a selling point of $3/kg for BDO and $35/kg for GBL, the total revenue will exceed $190M/yr, so it is strongly recommended the ECC pursue this opportunity.
Introduction
Project Justification
1,4 Butanediol is an important organic chemical used mainly as an intermediate for the production of Tetrahydrofuran (THF), Gamma-Butyrolactone (GBL) and Polybutylene Terephthalate (PBT). In 2011, the global market for BDO reached 1,725.0 kilo tons. Current projections suggest that worldwide BDO consumption will grow to 2550 kilo tons by 2017. The Technology Division of Evanston Chemicals has requested a preliminary design and economic evaluation on the feasibility of producing 1,4 Butanediol from a biomass derived succinic acid.
Technology Review
BDO can be produced by hydrogenation of succinic acid in the presence of a catalyst, at high temperatures and pressures (approximately 150-200°C and 20,000 kPa). However, to the best of the authors’ knowledge, there does not exist a means to produce only BDO. Instead, a mixture of THF, GBL, and BDO is produced. The choice of catalyst is the major factor in controlling yields and selectivity of a certain product. Current research suggests that bimetallic catalysts have a stronger activity and lead to higher conversion to BDO. TiO supported 2%Pd/X%Re catalyst and Carbon supported 2%Ru/4%Re catalyst both have been shown to be particularly effective. It is interesting to note that both catalysts contain Re. Research suggests that the synergy between Re and Pd/Ru leads to higher selectivity for BDO. Additionally, the chosen catalyst for this study was a 0.4% Fe, 1.9% Na, 2.66% Ag, 2.66% Pd, 10.0% Re on 1.5 mm carbon support. Taken from an ISP Investments patent from 2011, this catalyst has exceptionally high yields, upwards of 99.7% conversion of succinic acid, as well as high yields of BDO. In total, the percentage conversions of succinic acid to various products are: BDO: 85.51 wt % GBL: 2.04 wt % THF: 9.28 wt % Butanol: 2.87 wt % The lifetime of the catalyst is approximately 5 years. Furthermore, multiple reactor types have been discussed in the literature, including CSTRs and PFRs. However, the literature also shows that gas liquid induction reactors are the best for hydrogenation reactions in industry. They are preferred for their safety with respect to explosive gas phase reactants and complete utilization of the gas phase reactant; being especially useful for expensive inputs. These reactors use energy efficient impellers for gas induction and dispersion along with special recovery equipment to prevent waste of the gas phase, thus GLIR was chosen for this study.
Design Basis
The plant has been proposed to be built in Lake Providence, Louisiana. This is due primarily to its proximity to suppliers of succinic acid in the south east region, as well as close to distributors by way of the Mississippi River and the Gulf of Mexico. Additionally, the plant has been designed to have a capacity of approximately 50MM kg/yr of BDO production. This will require feeds of compressed hydrogen gas, and an aqueous feed of water and succinic acid, in a 50/50 wt % composition.
Technical Approach
The primary tool used to model the plant process was Aspen HYSYS. Because of the non-idealities involved in the NRTL HYSYS fluid package, an additional fluid package was created in Aspen. Additionally, the GLIR reactor chosen was modeled as a conversion reactor. Selectivity and yield of the reactor was modeled based off the specific data from the ISP Investments patent (discussed in Introduction: Technology Review). Although this provided very accurate modeling, the operating pressure and temperature were required to be held constant. This prevented any optimization within the reactor with respect to these operating conditions. The distillation columns were modeled as fractional distillation columns, with bottoms or distillate flow rate, and component recovery as active constraints. Additionally, Aspen Energy Analyzer was used to create the heat exchanger network, which can be seen in the Appendix. The economic analysis was performed in Aspen ICARUS.
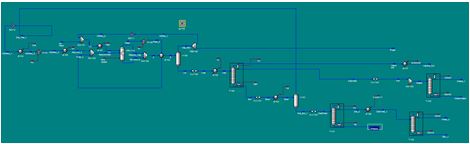
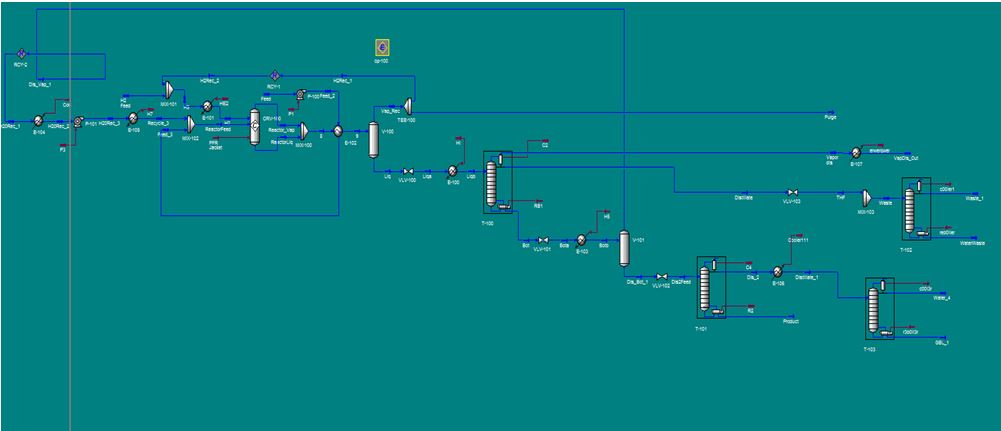
Figure 1. Screenshot of Aspen HYSYS process simulation.
As mentioned above, this plant will produce technical grade BDO and GBL, but the THF stream will be treated as waste. Originally, attempts were made to purify the THF stream in order to sell it; this process produces approximately 5M kg/yr, and at a selling point of ~$3.00/kg, this would generate an additional $15M/yr in revenue. However, the stream containing THF also contains water and n-butanol. As shown in Appendix XX, THF and water have a pressure dependent azeotrope; Figures XX1 and XX2 show that the azeotrope can be broken by pressurizing the stream after initial separation and atmospheric pressure. However, the presence of n-butanol makes separation of THF and water impossible; butanol and water have a tight azeotrope that cannot be broken simply by pressurizing the stream. Instead, absorption or ion exchange must be used to purify the stream. After extensive efforts were made to purify the THF, it was decided that the undertaking would not be profitable. In future iterations of this project, redoubled efforts would be made to force separation of THF to generate large revenues.
Process Description
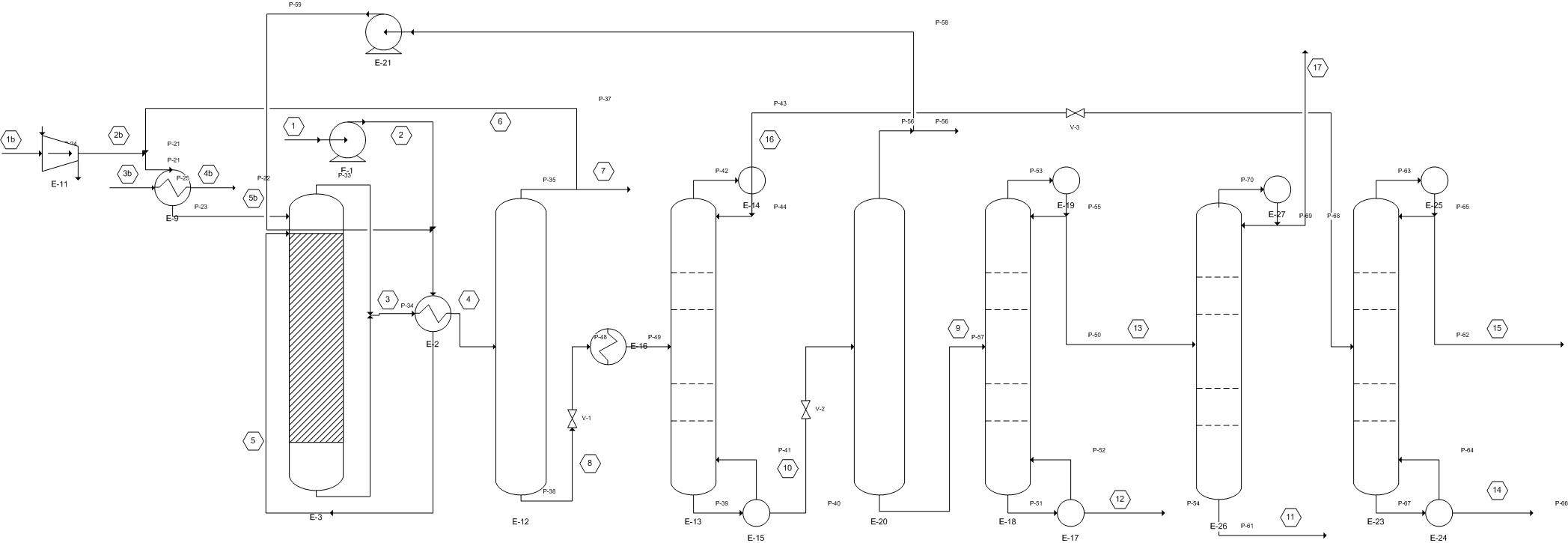
There are three main component of our process: pre-treatment, reaction and separation. See Appendix 15 for a detailed PFD of the process to reference.
Pre-treatment Phase
In the pretreatment phase of our process we bring all of the components to the correct conditions in order to be fed into the reactor. For example the 50% succinic acid in water feed in pumped to a pressure of 3250 psia and then heated to about 160oC, which are the operating conditions of the gas-liquid induction reactor used in the process. The operating temperature is relatively low in order to promote selectivity toward BDO and prevent other side reactions. At higher temperatures the hydrogenation of succinic acid continues past BDO and creates unwanted products. See Appendix 14 for the full reaction scheme of this process. At this point the feed can be mixed with a recycled steam coming off of our second gas-liquid separator, which is composed of water, BDO and BGL. It was advantageous to combine these two steams entering the reactor because the recycle stream saves excess water that was separated out before the final product and further dilutes the succinic acid concentration, for stabilization purposes, within the feed stream. Furthermore, the excess BDO that is filtered out and would be lost is recycled back into the system to improve the system recovery. Finally the GBL can be further reacted in the presence of hydrogen in order to form BDO, or once again recovered at the end of the process to be purified and sold rather than wasted. Of course the recycled stream must be pressurized and heated to the correct conditions as well, however, we first cool the recycle stream to form a liquid so that it can be pumped to the correct pressure of 3250 psia (rather than compressed as a gas, since compressors are much more expensive) and finally heated back to a temperature of 160oC. On the other side of the GLIR the hydrogen feed enters the process. Hydrogen purchased at 3000 psia is compressed to 3250 psia and then mixed with recycled hydrogen coming off the top of our first gas-liquid separator. The hydrogen from the top of the separator is split so that some is purged while the rest is recycled (still at high pressure) and mixed with the newly purchased hydrogen. It was necessary to recycle hydrogen because it is such an expensive input, and used in major excess to simply sending all of it out in the purge to a flare would be very wasteful. In future iterations of the project, the energy created from hydrogen burned off at a flare should be captured and used in the heat exchange network (explained later). The mixed streams, still at high pressure, are then heated to 160oC and fed into the reactor.
Reaction Phase
The reaction is to take place in a gas-liquid induction reactor, GLIR for short. The GLIR was chosen because it is best suited for multi-phase gas-liquid reactions. In particular the GLIR has special safety features to ensure worker’s safety under intense operating conditions, such as the high pressure used in our reactor, or when the reactant gas is highly combustible, as hydrogen is in our case. Additionally, GLIR’s are known for their complete utilization of the gas feed and great recovery specs with few losses, which is important for an expensive input feed. In our case, hydrogen can cost upwards of $7/kg and so this is a worthwhile investment. The GLIR will be a sort of fluidized bed reactor holding the catalyst with the liquid feed being pumped through downward as the gas bubbles through the liquid upward while an impeller stirs the mixture to increase contact times. Using a catalyst composed of a combination of Pd/Re/Ag/Na, typical for a hydrogenation process, high conversion of succinic acid and selectivity toward BDO can be achieved. Adding Fe to the catalyst has been shown to further increase conversion of succinic acid and selectivity toward BDO and so that will be done as well. Using a 0.4% Fe, 1.9% Na, 2.66% Ag, 2.66% Pd, 10% Re catalyst with the balance of the 1.5mm diameter carbon support, we are able to achieve 99.7% conversion of succinic acid with 85.5% selectivity to BDO, 2.04% selectivity to GBL, 9.3% selectivity to THF and the balance to other side products but namely butanol (but also possibly propanol and methane, etc.). The reactor operates isothermally and isobarically at 160oC and 3250 psia. Since the reaction is exothermic a cooling jacket will be placed on the GLIR in order to remove the excess heat generated. The jacket will contain a high heat capacity oil, namely Dowtherm A, that will then be put through a heat exchanger in order to maintain the oil at about 160oC at all times.
Separation Phase
The separation of the reactor effluent is the final part of our process. Since the effluent contains so many different components, many different reflux drums (gas-liquid separators) and fractionation columns are necessary. First, the effluent is cooled in a heat exchanger in order to allow for better separation of the hydrogen and liquid products in the reflux drum that follows where the hydrogen is taken out of the effluent mixture and either burned or recycled as described above. Next the remaining products and sent to a let-down valve to depressurize them and then reheated to promote better separation in the distillation column that follows which will separate out the main wastes (THF, water, butanol) from the main products (GBL, BDO). The first waste stream coming off of the top of this column is cooled and then sent to a waste tank as it is highly concentrated with butanol and THF. The other stream is further depressurized and sent to a distillation column to separate a stream highly concentrated in butanol and THF (as above) out, to be put into a waste container, from a stream that is >99% water which will be let out into the Mississippi River. The bottoms stream from the first fractionation column containing the main products is once again depressurized and heated to promote a better separation and then put into a new reflux drum. Here a stream high in water content is separated and recycled from a stream highly concentrated with the desired products. The non-recycled stream is sent to, yet another, distillation column which separates out our major product, 99.7% BDO in the bottoms from the GBL and water. The distillate stream is, once again, heated for better separation and sent to our last distillation column which separates out our other major product, >99% GBL, out of the bottoms from a water waste stream in the distillate which will also be let out into the Mississippi. Further iterations of the project could look at better ways to separate our products (using less columns/drums) or isolating other side-products to be resold (like THF). Additionally, other ways to handle side-product waste should be explored. Please see Appendix 6 for a closer look at our modeled HYSYS simulation.
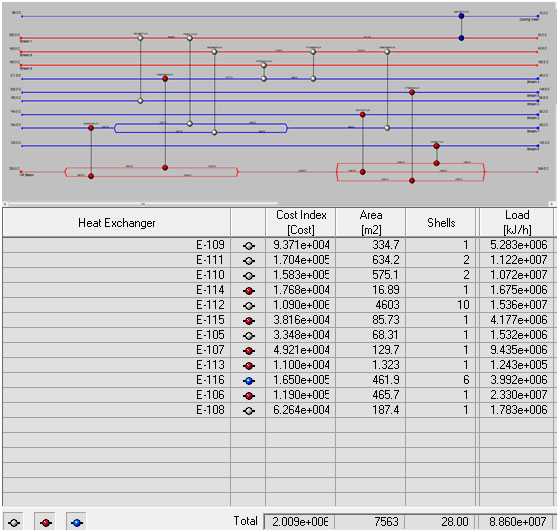
Heat Exchange Network (HEN)
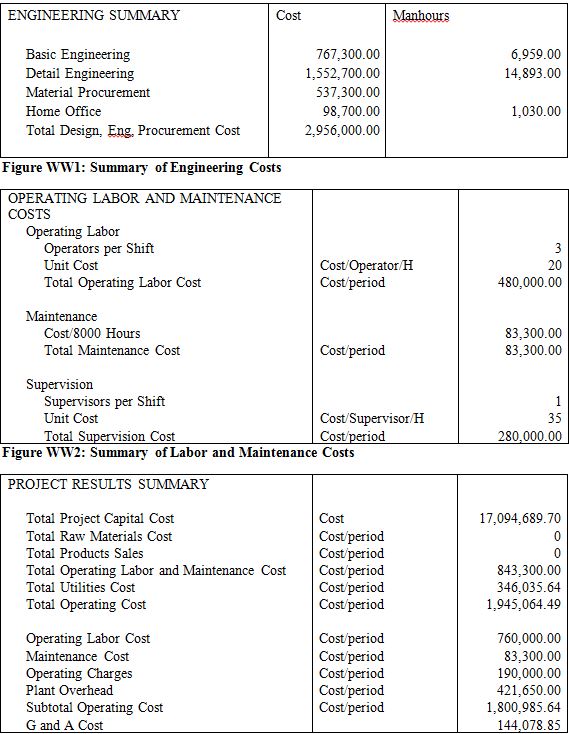
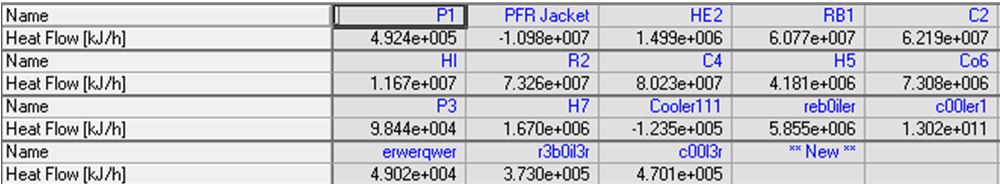
Since there are 9 streams that must be heated or cooled in our plant as well as a stream from the reactor jacket, a heat exchange network is necessary to minimize our total plant utilities in a heat exchange network (HEN). Using pinch point analysis it was determined that the best network contained 12 total heat exchangers with one stream split using a total of 3.87 x 107 kJ/h of hot utility and 3.99 x 106 kJ/h of total cold utility. These were necessary due to the complex nature of optimizing 10 different process streams needing temperature adjustment while avoiding temperature crosses, etc. Most of the streams exchange energy between different process streams while only one uses a cooling water and five use small amounts of high pressure steam, which account for the utilities needed. For more details of the HEN see Appendix 13. Further iterations of the project should consider adding the duties of the four reboilers and four condensers used on our distillation columns to the heat exchange network. As is, the network only considers the heat exchangers needed for process streams in order to minimize total utilities used within the process, however, a significant portion of our utilities is used in our distillation columns and thus future iterations could further reduce this number and make the plant more profitable by incorporating them into the network.
Process Flow Diagram & Flow Sheets
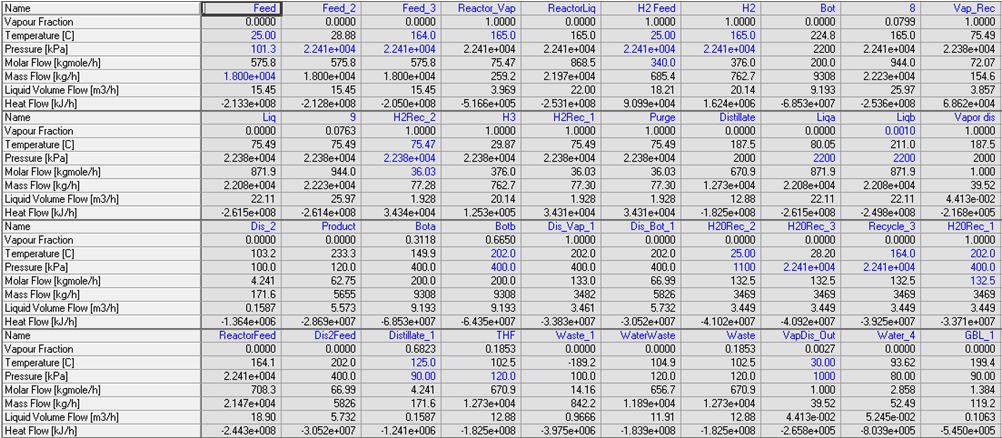
Below is listed a complete flow sheet, with data taken from Aspen HYSYS.
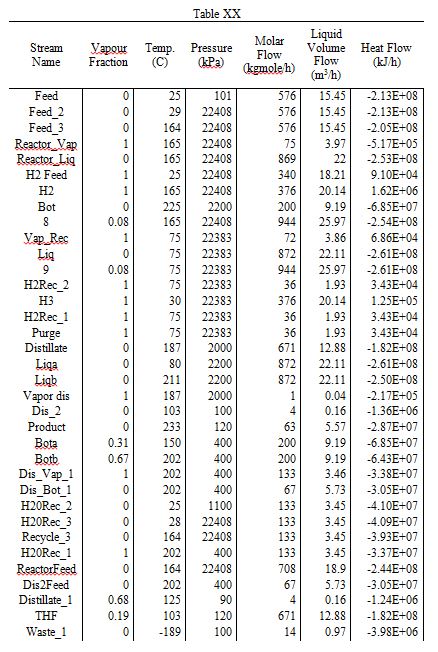
Table 1
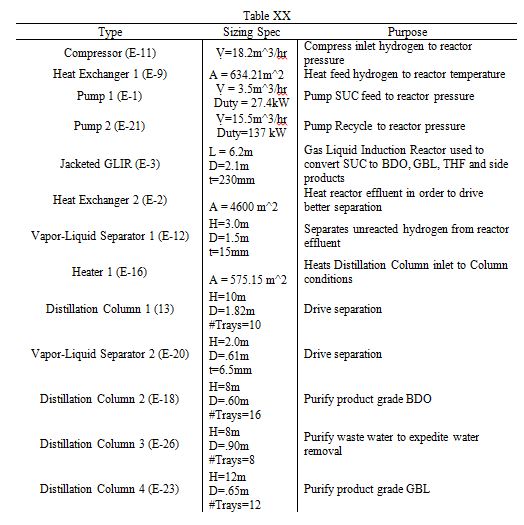
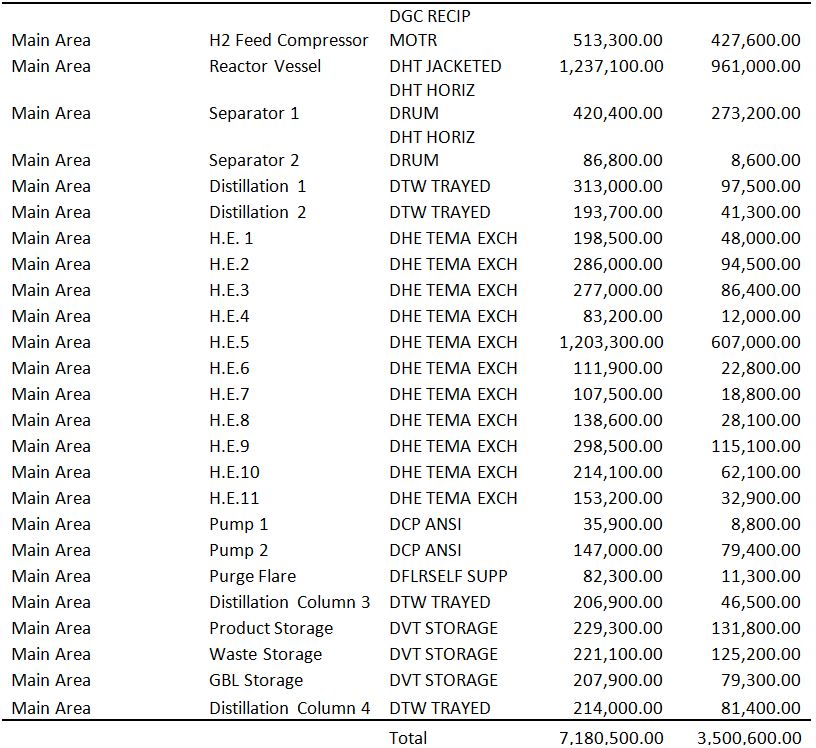
A detailed sizing list of the relevant components is listed below. Of particular interest are the distillation columns, which have heights between 8 and 12 meters. Furthermore, the reactor has a length of 6.2 meters and a diameter of 2.1 meters. A more complete table of sizing and sizing methodologies is listed in the Appendix.
Table 2
Economic Analysis
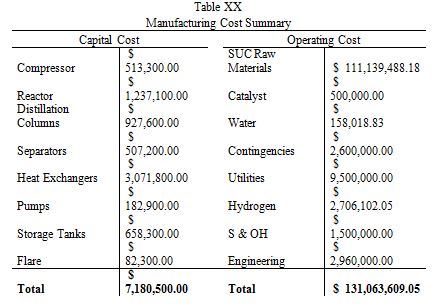
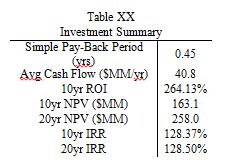
Table 3
Table 3 is a summary of the capital and operating costs required for the production of BDO and GBL. The expenditure on succinic acid clearly stands out; it makes up roughly 80% of the annual operating costs of this plant. The production plant consumes approximately 75.6M kg/yr of succinic acid, and at $1.47/kg, the costs add up quickly. Some other things to note from Table XX; the reactor is the most expensive singular piece of equipment; because it operates at exceedingly high pressure, it has extremely thick walls and is made of a high strength steel alloy. Additionally, the total cost of the heat exchangers is more than $3M; however, the production plant uses twelve heat exchangers, so the average individual cost comes out to a more reasonable $280,000. Lastly, this plant has a high annual utilities cost, mainly because of the 4 distillation columns and the large amount of cooling water necessary to keep the process running at the correct temperature. Below in Table 4 is a summary of the economic measures of return of this process.
Table 4
As shown clearly in Table 4, this plant has extremely encouraging economic potential, with an average yearly cash flow of $40.8M, a simple payback period of less than 6 months, and a breakeven point of approximately 9 months. These capital projections assume a 2 year construction time, 38% tax rate, 50% capacity in the first year of production, and a 7 year MACRS depreciation method. Revenue is split into two streams; main product (BDO) and by-product (GBL). The main product revenue is approximately $150M/yr, while the by-product revenue is $40M/yr, despite producing nearly 50 times less GBL than BDO. In future iterations of this design, it may be worthwhile to examine if it is more profitable to convert all of the succinic acid directly to GBL instead of producing the intermediary, BDO. If the economic returns seem astronomical, it is because they are; no economic investment actually produces these kinds of returns. The extremely favorable prediction could stem from several sources of error; supply and demand, bulk pricing, waste removal, and side reactions. The yearly supply of BDO is approximately 1M metric tons, meaning that this plant would produce 5% of the world’s supply. If the supply of BDO outpaced the demand, the selling price would fall, cutting into main product revenues. Additionally, the estimate of GBL selling price was based off a per kg price; if bulk pricing were used, the selling price of GBL would be between 40-50% lower than the price used. In addition to economic discontinuities, the problem of waste removal was never fully solved. Based on past estimates, waste removal could cost as must as $10M/year, further reducing the profitability of this plant. Lastly, the process of BDO synthesis was simplified for modelling purposes. In actual production, many side reactions would occur; driving down conversion and increasing separation costs.
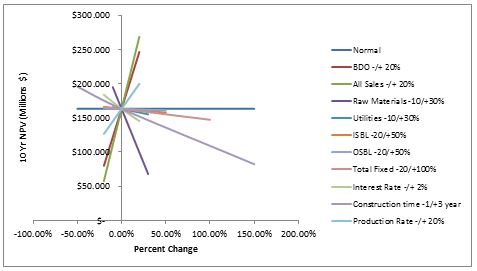
A sensitivity analysis was performed on all important variables.
Figure 1: Sensitivity analysis
Figure 1 shows that the profitability of the plant (based on the 10 year NPV), is most sensitive to the selling price of BDO and GLB, the cost of raw materials, and the construction time. It is least sensitive to changes in the utilities cost and fixed capital costs. It is clear from the figure that this undertaking is profitable in the face of any singular change. However, if several of these variables react unfavorably to the coming economic climate, the plant could potentially make very little money. For example, if the construction time doubles (which happens frequently) and the selling prices of BDO and GBL are driven down by market saturation, the plant could have a 10 year NPV falls to approximately 0.
Conclusion
To conclude, this project is highly feasible, and our company stands to make a very high profit from the construction of this plant. Based on a two year construction time, a 38% tax rate, and 50% production over the first year, our simple payback period would be 0.45 years, with yearly revenue of over $190M. We strongly recommend Evanston Chemical to move forward with this facility. However, there are key next steps to be considered. First, a more detailed reaction scheme for the reactor is required. This scheme must be able to handle variable operating pressure and temperature, as well as contain a more detailed and complete incorporation of side reactions. Namely, the reaction of GBL to BDO is a necessary consideration. Moreover, a fully life cycle analysis and environmental health and safety analysis will be required. The removal of waste will also need to be addressed. Additionally, profits may be increased by performing a plant wide optimization project. Also, the assumption of a 5 year catalyst lifetime will need to be addressed in more detail, as the high cost of this component will greatly affect the bottom line. Another factor which may affect the overall profitability is the flocculating prices of rare metals, which make up the catalyst. Also, the market price of these commodity chemicals will almost certainly be affected by a 10% influx of global supply. Finally, while we have planned on also selling GBL, looking into purifying the THF will also be profitable.
Appendices
Appendix 1
Sizing and construction information.
Appendix 2
Equipment Costs
Appendix 3
Economic Summary
Appendix 4
Energy Stream Summary
Appendix 5
HYSYS Simulation
Appendix 6
Stream Summary Table
Appendix 7
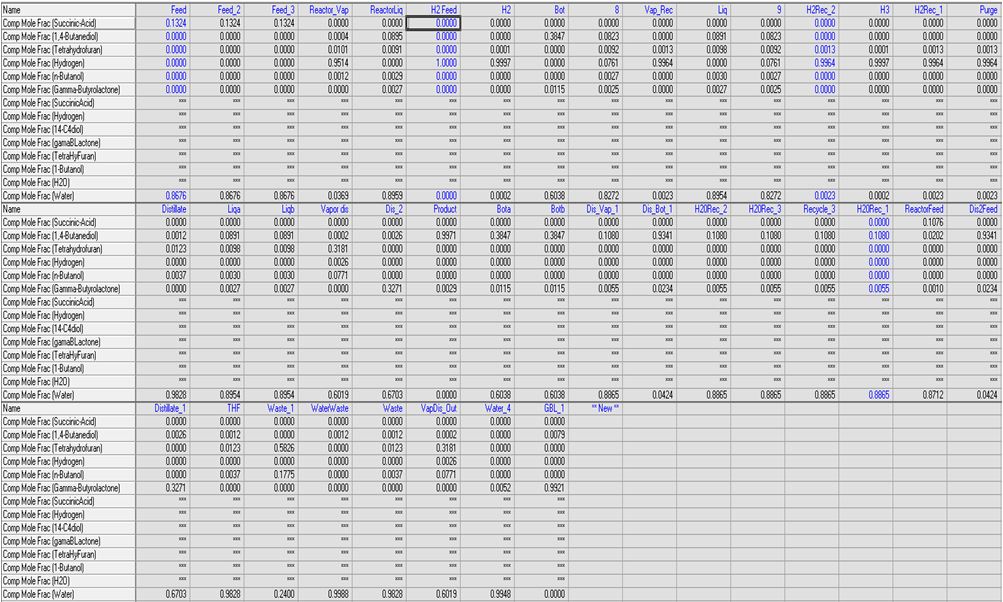
Summary of HYSYS stream compositions.
Appendix 8
Heat exchanger network.
Appendix 9
Reaction mechanism summary
Appendix 10
Process Flow Diagram